NEET 2025 Chemistry Question Paper with Solutions PDF is available for download. NEET 2025 Chemistry Question Paper comprises 45 MCQs. NEET 2025 has been conducted on May 4, 2025. You can download NEET 2025 Chemistry question paper with answer key and solutions PDF using the links given below.
Check:
NEET 2025 Chemistry Question Paper with Solutions PDF
| NEET 2025 Chemistry Question Paper with Answer Key | Download PDF | Check Solution |

Given below are two statements:
Statement I: Ferromagnetism is considered as an extreme form of paramagnetism.
Statement II: The number of unpaired electrons in a Cr2+ ion (Z = 24) is the same as that of a Nd3+ ion (Z = 60).
In the light of the above statements, choose the correct answer from the options given below:
View Solution
For the reaction A(g) \(\rightleftharpoons\) 2B(g), the backward reaction rate constant is higher than the forward reaction rate constant by a factor of 2500, at 1000 K.
[Given: R = 0.0831 atm {–1} K{–1}]
Kp for the reaction at 1000 K is:
View Solution
Total number of possible isomers (both structural as well as stereoisomers) of cyclic ethers of molecular formula C4H8O is:
View Solution
Given below are two statements:
Statement I: A hypothetical diatomic molecule with bond order zero is quite stable.
Statement II: As bond order increases, the bond length increases.
In the light of the above statements, choose the most appropriate answer from the options given below:
View Solution
Identify the suitable reagent for the following conversion:

View Solution
The major product of the following reaction is:

(i) CH3MgBr (excess)
(ii) H+/H2O
View Solution
If the molar conductivity (\(\Lambda_m\)) of a 0.050 mol L–1 solution of a monobasic weak acid is 90 S cm2 mol–1, its extent (degree) of dissociation will be:
[Assume: \(\Lambda^0 = 349.6\) S cm2 mol–1 and \(\Lambda^0_{acid} = 50.4\) S cm2 mol–1]
View Solution
Which one of the following reactions does NOT belong to “Lassaigne's test”?

View Solution
The correct order of decreasing acidity of the following aliphatic acids is:

View Solution
Match List I with List II:

Choose the correct answer from the options given below:
View Solution
Out of the following complex compounds, which of the compound will be having the minimum conductance in solution?
View Solution
Sugar ‘X’:
A. is found in honey.
B. is a keto sugar.
C. exists in \(\alpha\) and \(\beta\) anomeric forms.
D. is laevorotatory.
‘X’ is:
View Solution
How many products (including stereoisomers) are expected from monochlorination of the following compound?
CH3-CH-CH2-CH3
View Solution
Which one of the following compounds can exist as cis-trans isomers?
View Solution
Which one of the following reactions does NOT give benzene as the product?
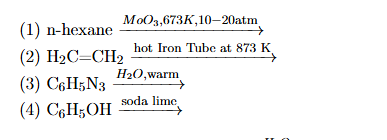
View Solution
Phosphoric acid ionizes in three steps with their ionization constant values \( K_1 \), \( K_2 \), and \( K_3 \), respectively, while \( K \) is the overall ionization constant.
Which of the following statements are true?
View Solution
Among the following, choose the ones with an equal number of atoms.
A. 212 g of Na2CO3 (s) [molar mass = 106 g]
B. 248 g of NaNO3 (s) [molar mass = 62 g]
C. 240 g of NaOH (s) [molar mass = 40 g]
D. 12 g of H2 (g) [molar mass = 2 g]
E. 220 g of CO2 (g) [molar mass = 44 g]
Choose the correct answer from the options given below:
View Solution
Given below are two statements:
Statement I: Like nitrogen that can form ammonia, arsenic can form arsine.
Statement II: Antimony cannot form antimony pentoxide.
In the light of the above statements, choose the most appropriate answer from the options given below:
View Solution
Dalton’s Atomic Theory could not explain which of the following?
View Solution
The correct order of decreasing basic strength of the given amines is:
View Solution
Which of the following statements are true?
A. Unlike Ga that has a very high melting point, Cs has a very low melting point.
B. On Pauling scale, the electronegativity values of N and C are not the same.
C. Ar, K+, Cl–, Ca2+ and S2– are all isoelectronic species.
D. The correct order of the first ionization enthalpies of Na, Mg, Al, and Si is Si > Al > Mg > Na.
E. The atomic radius of Cs is greater than that of Li and Rb.
Choose the correct answer from the options given below:
View Solution
Match List I with List II:

Choose the correct answer from the options given below:
View Solution
The standard heat of formation, in kcal/mol, of Ba2+ is:
Given: Standard heat of formation of SO₄2–(aq) = -216 kcal/mol, standard heat of crystallization of BaSO₄(s) = -4.5 kcal/mol, standard heat of formation of BaSO₄(s) = -349 kcal/mol.
View Solution
Among the given compounds I-III, the correct order of bond dissociation energy of the C-H bond marked with is:
View Solution
Predict the major product \( P \) in the following sequence of reactions:
(i) HBr, benzoyl peroxide
(ii) KCN
(iii) Na(Hg), C2H5OH
View Solution
Match List I with List II:

Choose the correct answer from the options given below:
View Solution
Energy and radius of first Bohr orbit of He+ and Li2+ are:
Given: \( R_H = 2.18 \times 10^{-18} \, J, a_0 = 52.9 \, pm \)
View Solution
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): I– undergoes SN2 reaction faster than Cl–.
Reason (R): Iodine is a better leaving group because of its large size.
In the light of the above statements, choose the correct answer from the options given below:
View Solution
If the half-life (\( t_{1/2} \)) for a first-order reaction is 1 minute, then the time required for 99.9% completion of the reaction is closest to:
View Solution
Which of the following aqueous solutions will exhibit the highest boiling point?
View Solution
Higher yield of NO in the reaction
N2 (g) + O2 (g) → 2NO (g)
can be obtained at [\( \Delta H \) of the reaction = +180.7 kJ mol\(^{-1}\)]
View Solution
Match List I with List II:

Choose the correct answer from the options given below:
View Solution
The ratio of the wavelengths of the light absorbed by a Hydrogen atom when it undergoes n = 2 → n = 3 and n = 4 → n = 6 transitions, respectively, is:
View Solution
The correct order of the wavelength of light absorbed by the following complexes is:
A. [ Co(NH3)6]3+
B. [ Co(CN)6]3–
C. [ Cu(H2O)4]2+
D. [ Ti(H2O)6]3+
Choose the correct answer from the options given below:
View Solution
Match List I with List II:

Choose the correct answer from the options given below:
View Solution
If the rate constant of a reaction is 0.03 s\(^{-1}\), how much time does it take for a 7.2 mol L\(^{-1}\) concentration of the reactant to get reduced to 0.9 mol L\(^{-1}\)?
(Given: log 2 = 0.301)
View Solution
Which among the following electronic configurations belong to main group elements?
A. [Ne]3s1
B. [Ar]3d34s2
C. [Kr]4d53s25p5
D. [Ar]3d104s1
E. [Rn]5f76d27s2
Choose the correct answer from the options given below:
View Solution
Match List I with List II:

Choose the correct answer from the options given below:
View Solution
5 moles of liquid X and 10 moles of liquid Y make a solution having a vapor pressure of 70 torr. The vapor pressures of pure X and Y are 63 torr and 78 torr, respectively. Which of the following is true regarding the described solution?
View Solution
C(s) + 2H\(_2\)(g) \(\rightarrow\) CH\(_4\)(g); \(\Delta H = -74.8 \, kJ mol^{-1}\)
Which of the following diagrams gives an accurate representation of the above reaction?
Which one of the following compounds does not decolorize bromine water?
View Solution
Consider the following compounds:
\text{K\(_2\)O\(_2\), H\(_2\)O\(_2\), and H\(_2\)SO\(_4\)
The oxidation states of the underlined elements in them are, respectively:
View Solution
Given below are two statements:
Statement I: Benzendiazonium salt is prepared by the reaction of aniline with nitrous acid at 273-278 K. It decomposes easily in the dry state.
Statement II: Insertion of iodine into the benzene ring is difficult and hence iodobenzene is prepared through the reaction of benzendiazonium salt with KI.
In the light of the above statements, choose the most appropriate answer from the options given below:
View Solution
Which of the following are paramagnetic?
A. [NiCl\(_4\)]\(^{2-}\)
B. Ni(CO)\(_4\)
C. [Ni(CN)\(_4\)]\(^{2-}\)
D. [Ni(H\(_2\)O)\(_6\)]\(^{2+}\)
E. Ni(PPh\(_3\))\(_4\)
Choose the correct answer from the options given below:
View Solution




































Comments