CBSE Class 10 Science Question Paper 2024 PDF (Set 1 - 31/4/1) is available for download here. CBSE conducted the Science exam on March 2, 2024, from 10:30 AM to 1:30 PM. The total marks for the theory paper are 80. The question paper contains 20% MCQ-based questions, 40% competency-based questions, and 40% short and long answer type questions.
CBSE Class 10 Science Question Paper 2024 (Set 1 - 31/4/1) with Answer Key
| CBSE Class 10 Science Question Paper 2024 (Set 1 - 31/4/1) with Answer Key | Check Solutions |
CBSE Science Question Paper (Set 1 – 31/4/1) 2024 Solution
| Question | Answer | Detailed Solution |
|---|---|---|
| 1. Zn + 2CH3COOH → (CH3COO)2Zn + H2 The above reaction is a: (A) Decomposition reaction (B) Displacement reaction (C) Double displacement reaction (D) Combination reaction |
(B) Displacement reaction | In this reaction, zinc displaces hydrogen from acetic acid to form zinc acetate and hydrogen gas. This is a characteristic of a displacement reaction, where a more reactive element replaces a less reactive element from its compound. |
| 2. An aqueous solution of a salt turns blue litmus to red. The salt could be the one obtained by the reaction of: (A) HNO3 and NaOH (B) H2SO4 and KOH (C) CH3COOH and NaOH (D) HCl and NH4OH |
(D) HCl and NH4OH | A salt that turns blue litmus paper to red is acidic in nature. HCl reacts with NH4OH to form NH4Cl, which is acidic. |
| 3. Four solutions, namely glucose, alcohol, hydrochloric acid, and sulphuric acid, are connected one by one in an electric circuit with a bulb. The solutions in which the bulb will glow are: (A) Glucose and alcohol (B) Alcohol and hydrochloric acid (C) Glucose and sulphuric acid (D) Hydrochloric acid and sulphuric acid |
(D) Hydrochloric acid and sulphuric acid | Hydrochloric acid and sulphuric acid ionize completely in water, conducting electricity. Glucose and alcohol do not ionize and hence do not conduct electricity. |
| 4. The metals which are found in both free state as well as combined state are: (A) Gold and platinum (B) Platinum and silver (C) Copper and silver (D) Gold and silver |
(C) Copper and silver | Copper and silver are found in the free state due to their low reactivity and in combined forms like copper sulphide and silver chloride. |
| 5. The number of single and double bonds present in a molecule of benzene (C6H6) respectively, are: (A) 6 and 6 (B) 9 and 3 (C) 3 and 9 (D) 3 and 3 |
(B) 9 and 3 | Benzene has 9 single bonds (6 C-H and 3 C-C) and 3 double bonds (C=C within the ring). |
| 6. In human beings, when the process of digestion is completed, the (i) proteins, (ii) carbohydrates, and (iii) fats are finally converted into: (A) (i) Amino acids, (ii) glucose, and (iii) fatty acids (B) (i) Amino acids, (ii) glucose, (iii) fatty acids and glycerol (C) (i) Glucose, (ii) fatty acids and glycerol, and (iii) amino acids (D) (i) Sugars, (ii) amino acids, and (iii) fatty acids and glycerol |
(B) (i) Amino acids, (ii) glucose, (iii) fatty acids and glycerol | Proteins are converted to amino acids, carbohydrates to glucose, and fats to fatty acids and glycerol during digestion. |
| 7. A plant growth inhibitor hormone which causes wilting of leaves is called: (A) Auxin (B) Cytokinin (C) Abscisic acid (D) Gibberellin |
(C) Abscisic acid | Abscisic acid is a plant hormone that promotes wilting, dormancy, and inhibits growth. It is also known as the stress hormone. |
| 8. The plants that can be raised by the method of vegetative propagation are: (A) Sugarcane, roses, grapes (B) Sugarcane, mustard, potato (C) Banana, orange, mustard (D) Papaya, mustard, potato |
(A) Sugarcane, roses, grapes | Vegetative propagation involves reproduction using parts like stems, roots, or leaves. Sugarcane, roses, and grapes propagate this way. |
| 9. The part of seed which is a source of food during germination of seed is: (A) Cotyledon (B) Radicle (C) Plumule (D) Embryo |
(A) Cotyledon | The cotyledon stores food for the developing embryo, providing nutrients during germination until the seedling can photosynthesize. |
| 10. A zygote is formed by the fusion of a male gamete and a female gamete. The number of chromosomes in the zygote of a human is: (A) 23 (B) 44 (C) 46 (D) 92 |
(C) 46 | In humans, each gamete contains 23 chromosomes. The fusion of a male and female gamete forms a zygote with 46 chromosomes (23 pairs). |
| 11. Absolute refractive index of glass and water is 3/2 and 4/3 respectively. If the speed of light in glass is 2 × 108 m/s, the speed of light in water is: (A) 9/4 × 108 m/s (B) 5/2 × 108 m/s (C) 7/3 × 108 m/s (D) 16/9 × 108 m/s |
(A) 9/4 × 108 m/s | The speed of light in a medium is given by ( v = c / n), where ( c = 3 × 108 m/s ). For water ( n = 4/3 ): ( v = (3 × 108) × (3/4) = 9/4 × 108 m/s ). |
| 12. When a beam of white light passes through a region having very fine dust particles, the colour of light mainly scattered in that region is: (A) Red (B) Orange (C) Blue (D) Yellow |
(C) Blue | According to Rayleigh's scattering law, shorter wavelengths like blue are scattered more than longer wavelengths, making the scattered light appear blue. |
| 13. Consider the following combinations of resistors: Combination I: Two 2 Ω resistors in parallel Combination II: Three 3 Ω resistors in parallel Combination III: Resistors arranged with 2 Ω and 4 Ω in series and parallel with 3 Ω resistors Combination IV: Two 0.5 Ω resistors in parallel The combinations having equivalent resistance 1 Ω is/are: (A) I and IV (B) Only IV (C) I and II (D) I, II and III |
(C) I and II | Combination I: ( 1/R = 1/2 + 1/2 = 2/2 ), so ( R = 1 Ω ). Combination II: ( 1/R = 1/3 + 1/3 + 1/3 = 3/3 ), so ( R = 1 Ω ). Both satisfy ( R = 1 Ω ). |
| 14. An electric iron of resistance 20 Ω draws a current of 5 A. The heat developed in the iron in 30 seconds is: (A) 15000 J (B) 6000 J (C) 1500 J (D) 3000 J |
(A) 15000 J | Heat developed: \( H = I^2Rt \). Substituting \( I = 5 A \), \( R = 20 Ω \), \( t = 30 s \): \( H = (5)^2 × 20 × 30 = 25 × 20 × 30 = 15000 J \). |
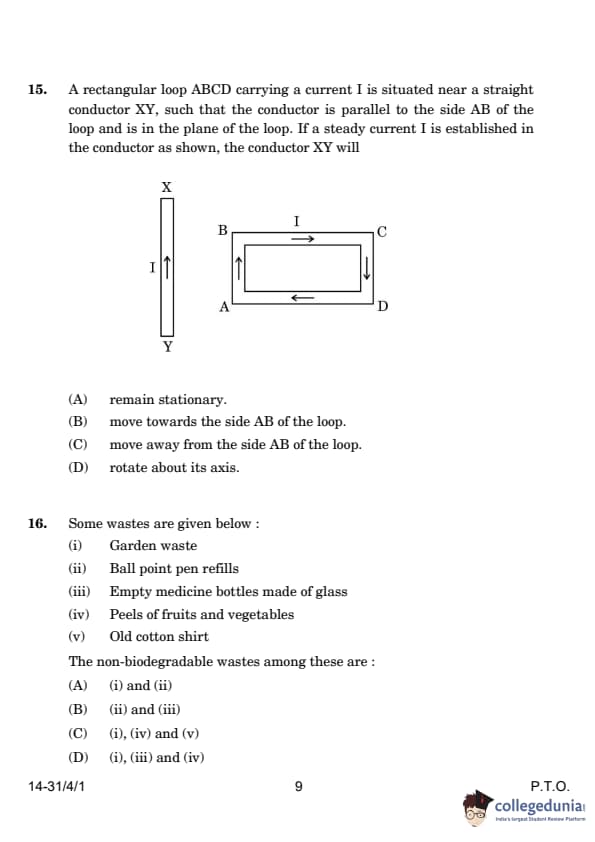
| 15. A rectangular loop carrying current I is situated near a straight conductor XY. If a steady current I is established in the conductor, the conductor will: (A) Remain stationary (B) Move towards the side AB of the loop (C) Move away from the side AB of the loop (D) Rotate about its axis |
(B) Move towards the side AB of the loop | Currents in the same direction attract each other due to the interaction of their magnetic fields. Hence, the conductor moves towards side AB. |
| 16. Some wastes are given below: (i) Garden waste (ii) Ballpoint pen refills (iii) Empty glass medicine bottles (iv) Peels of fruits and vegetables (v) Old cotton shirt The non-biodegradable wastes among these are: (A) (i) and (ii) (B) (ii) and (iii) (C) (i), (iv) and (v) (D) (i), (iii) and (iv) |
(B) (ii) and (iii) | Non-biodegradable wastes include ballpoint pen refills and empty glass medicine bottles as they do not decompose naturally. |
| 17. Assertion (A): The extraction of metals from their sulphide ores cannot take place without roasting the ore. Reason (R): Roasting converts sulphide ores directly into metals. (A) Both A and R are true, and R is the correct explanation of A (B) Both A and R are true, but R is not the correct explanation of A (C) A is true, but R is false (D) A is false, but R is true |
(C) A is true, but R is false | Roasting converts sulphide ores into oxides, not directly into metals. Oxides are then reduced to extract metals. |
| 18. Assertion (A): In the human heart, ventricles have thicker muscular walls than atria. Reason (R): Ventricles have to pump the blood into various organs. (A) Both A and R are true, and R is the correct explanation of A (B) Both A and R are true, but R is not the correct explanation of A (C) A is true, but R is false (D) A is false, but R is true |
(A) Both A and R are true, and R is the correct explanation of A | The ventricles have thicker walls because they pump blood with greater force to organs, unlike the atria which only pump to ventricles. |
| 19. Assertion (A): Magnetic field lines never intersect each other. Reason (R): If they intersect, the compass needle would point towards two directions, which is not possible. (A) Both A and R are true, and R is the correct explanation of A (B) Both A and R are true, but R is not the correct explanation of A (C) A is true, but R is false (D) A is false, but R is true |
(A) Both A and R are true, and R is the correct explanation of A | Magnetic field lines do not intersect because at any point, the magnetic field has only one direction. Intersecting lines would imply multiple directions. |
| 20. Assertion (A): Oxygen is essential for all aerobic forms of life. Reason (R): Free oxygen atoms combine with molecular oxygen to form ozone. (A) Both A and R are true, and R is the correct explanation of A (B) Both A and R are true, but R is not the correct explanation of A (C) A is true, but R is false (D) A is false, but R is true |
(B) Both A and R are true, but R is not the correct explanation of A | Oxygen is required for aerobic respiration, but ozone formation is unrelated to its role in sustaining life. |
| 21. (a) 1 gram of solid sodium chloride was taken in a clean and dry test tube and concentrated sulphuric acid was added to it. (i) Name the gas evolved in the reaction. (ii) What will be observed when this gas is tested with (I) dry, and (II) wet blue litmus paper? Write your conclusion about the nature of this gas. |
(a) (i) Hydrogen chloride (HCl). (ii) (I) No change (dry litmus); (II) Turns red (wet litmus). Conclusion: The gas is acidic in nature. |
(a) (i) Hydrogen chloride gas (HCl) is evolved when NaCl reacts with concentrated H2SO4. (ii) Observations: - (I) Dry blue litmus: No change. - (II) Wet blue litmus: Turns red due to the acidic nature of HCl. |
| OR 21.(b) Some metals react with acids to produce salt and hydrogen gas. Illustrate it with an example. How will you test the presence of this gas? | (b) Example: Zn + 2HCl → ZnCl2 + H2. Test: Burns with a pop sound. |
(b) Example: Zinc reacts with hydrochloric acid to form zinc chloride and hydrogen gas. Test: A burning splint produces a pop sound in the presence of hydrogen gas. |
| 22. Distinguish between a saturated and an unsaturated hydrocarbon by flame test. List the products of the combustion reaction of a saturated hydrocarbon. | Saturated: Clean flame. Unsaturated: Sooty flame. Products: CO2, H2O, energy. |
Saturated hydrocarbons burn with a clean, blue flame due to complete combustion, while unsaturated hydrocarbons burn with a yellow, sooty flame due to incomplete combustion. Combustion products of saturated hydrocarbons: Carbon dioxide (CO2), water (H2O), and energy. |
| 23. (a) We need to water the soil in plants on a regular basis. But it ultimately reaches the leaves of the plant. Explain how this takes place. | (a) Root absorption, xylem transport, transpiration pull. (b) Holozoic nutrition; ingestion by pseudopodia. |
(a) Water is absorbed by root hairs through osmosis and transported upward via xylem. Transpiration pull drives water to the leaves. (b) Amoeba exhibits holozoic nutrition. It ingests food using pseudopodia to form a food vacuole, digests it with enzymes, absorbs nutrients, and expels waste. |
| OR 23.(b) Name the type of nutrition exhibited by Amoeba. Explain how food is taken in and digested by this organism. | ||
| 24. The survival of a species is promoted through creation of variations. Illustrate with an example. | Example: Peppered moths during industrialization. | Variations help species adapt to changing environments. For example, during the Industrial Revolution, dark-colored moths survived better due to their ability to blend with soot-darkened trees, promoting their survival. |
| 25. A person suffering from presbyopia needs bifocal lenses. If he needs two lenses of power −4.0 dioptre and +2.0 dioptre, which one of these two lenses is for the correction of distant vision and what is its focal length? | Correction: −4.0 D for distant vision. Focal length: −25 cm. |
A concave lens of power −4.0 dioptre is used to correct distant vision. Focal length \( f = 100/P = 100/−4 = −25 \) cm (negative sign indicates a concave lens). |
| 26. Two wires A and B of the same material, having lengths and diameters 0.2 mm and 0.3 mm respectively, are connected one by one in a circuit. Which one of these two wires will offer more resistance to the flow of current in the circuit? Justify your answer. | Wire A offers more resistance. | Resistance \( R \propto 1/A \), where \( A = πd^2/4 \). For wire A (smaller diameter), the cross-sectional area is less, resulting in higher resistance compared to wire B. |
| 27. Write the common name and chemical name of CaSO4·1/2H2O. Write the method of its preparation. Give the chemical equation for the reaction when it reacts with water. | Common Name: Plaster of Paris. Chemical Name: Calcium sulphate hemihydrate. |
Preparation: Heat gypsum (CaSO4·2H2O) to 373 K to remove water. Equation: CaSO4·2H2O → CaSO4·1/2H2O + 1.5H2O. Reaction with water: CaSO4·1/2H2O + 1.5H2O → CaSO4·2H2O. |
| 28. Why is the conversion of ethanol to ethanoic acid an oxidation reaction? Name the oxidizing agent used in this conversion. Write the chemical equation for the reaction. How is this reaction different from the combustion of ethanol? | Oxidation adds oxygen. Oxidizing Agent: KMnO4. Reaction: C2H5OH → CH3COOH. |
Oxidation involves adding oxygen to ethanol, forming ethanoic acid. In combustion, ethanol reacts with O2 to produce CO2 and H2O. Oxidizing agent: KMnO4 or K2Cr2O7. Equation: C2H5OH + [O] → CH3COOH + H2O. |
| 29. (a) Explain with a labeled diagram the process of reproduction in Hydra by budding. Name the cells used for reproduction in this process. | (a) Budding with regenerative cells. | (a) Hydra reproduces by budding using regenerative cells. A small bud forms, grows, and detaches to become an independent organism. |
| OR 29.(b) List two roles of each of the following: (i) Seminal vesicles and prostate gland (ii) Oviduct (iii) Testis |
(b) (i) Seminal vesicles: Nutrients. Prostate gland: Neutralizes acidity. (ii) Oviduct: Fertilization site. (iii) Testis: Sperm and testosterone. |
(b) (i) Seminal vesicles secrete fructose-rich fluid; the prostate gland secretes alkaline fluid to neutralize acidity. (ii) The oviduct is the site for fertilization and helps transport the zygote to the uterus. (iii) The testis produces sperms and testosterone for secondary sexual characteristics. |
| 30. List two differences between dominant and recessive traits. What percentage of pea plants in the F2 generation had yellow seeds in Mendel’s cross between yellow (YY) and green (yy) seeds? | Dominant: Expressed in heterozygous. Recessive: Only in homozygous. Yellow seeds: 75%. |
Dominant traits are expressed in heterozygous conditions and mask recessive traits. Recessive traits are expressed only in homozygous conditions. In Mendel’s cross, the F2 generation had a 3:1 ratio; 75% had yellow seeds. |
| 31. Define the term "power of accommodation" of the human eye. What happens to the image distance in the eye when we increase the distance of an object from the eye? Name and explain the role of the part of the human eye responsible for it in this case. | Power of accommodation: Adjusting focal length. Image distance: Constant. Responsible part: Ciliary muscles. |
Power of accommodation refers to the ability of the eye lens to adjust its focal length to focus on objects at varying distances. When the distance of the object increases, the image distance remains constant as the image is always formed on the retina. The ciliary muscles adjust the lens curvature, allowing clear vision for both distant and nearby objects. |
| 32. A student fixes a sheet of white paper on a drawing board, places a bar magnet in the center, and sprinkles iron filings around it. Then he taps the board gently. Answer the following: (a) Why do iron filings arrange in a particular pattern? (b) What does the crowding of iron filings at the poles indicate? (c) What do the lines along which the iron filings align represent? (d) If the student places a cardboard horizontally in a current-carrying solenoid and repeats the activity, what pattern would the filings form? |
(a) Magnetic field lines. (b) Strongest at poles. (c) Magnetic field lines. (d) Parallel inside, curved outside. |
(a) Iron filings align along magnetic field lines created by the bar magnet. (b) Crowding at poles indicates the magnetic field is strongest at the poles. (c) The lines represent magnetic field lines, indicating the direction and strength of the field. (d) Inside a solenoid, filings form parallel straight lines indicating a uniform magnetic field; outside, they form curved patterns resembling a bar magnet. |
| 33. Study the food chains below and answer: (a) Name the type of ecosystems in food chains (b) and (c). (b) Why are producers the first trophic level in all food chains? What percentage of solar energy do they capture for photosynthesis? (c) Why are the arrows in food chains unidirectional? |
(a) (b) Desert and aquatic. (b) Autotrophs; 1%. (c) Energy flows one way. |
(a) (b) Desert ecosystem for chain (b) and aquatic ecosystem for chain (c). (b) Producers are autotrophs that convert solar energy into chemical energy. They capture 1% of solar energy for photosynthesis. (c) Arrows are unidirectional because energy flows from producers to higher trophic levels and cannot flow back, as energy is lost as heat at each level. |
| 34. (a) What is a chemical reaction? Describe one activity each to show that a chemical change has occurred involving (i) a change in color and (ii) a change in temperature. |
(a) Change of color: CuSO4 + Fe. Change in temperature: CaO + H2O. |
(a) A chemical reaction transforms substances into new products. - Change in color: Add iron nails to copper sulfate solution (blue to green color change). - Change in temperature: Add water to quicklime (heat release). |
| OR 34.(b) Define a decomposition reaction. Give examples of (i) electrolysis of water and (ii) blackening of silver bromide, specifying the energy type involved in each. |
(b) Breakdown of a compound. Energy: Electric and light. |
(b) Decomposition reaction involves breaking a compound into simpler substances. - Electrolysis of water: Energy type - electrical. - Blackening of AgBr: Energy type - light. |
| 35. (a)(i)Define a reflex arc. Why have reflex arcs evolved in animals? Trace the sequence of events when touching a hot object. | (a) Reflex arc: Quick response pathway. Events: Sensory to spinal cord to motor nerve. |
(a) Reflex arc is the neural pathway for an automatic response to stimuli, evolved for quick protection. Sequence: Stimulus → sensory nerve → spinal cord → motor nerve → action. |
| 35. (a) (ii) Name the part of the nervous system which helps in communication between the central nervous system and other parts of the body. What are the two components of this system? | Peripheral Nervous System (PNS). Components: Somatic and Autonomic Nervous System. |
The Peripheral Nervous System (PNS) connects the central nervous system (CNS) to other parts of the body, enabling communication and coordination. The PNS has two components: - Somatic Nervous System: Controls voluntary actions like muscle movements. - Autonomic Nervous System: Regulates involuntary actions such as heartbeat and digestion. |
| OR 35.(b) Leaves of "chhui-mui" plant fold upon touch. Name the stimulus, explain the cause, and state if growth is involved. Define geotropism and provide examples of positive and negative geotropism. | (b) Stimulus: Touch. Geotropism: Roots positive; shoots negative. |
(b) Touch stimulus causes rapid movement in "chhui-mui" due to turgor pressure changes. No growth is involved. Geotropism: Directional growth due to gravity. Roots show positive geotropism, while shoots exhibit negative geotropism. |
| 35. (b) (ii) Define geotropism in plants. What is meant by positive and negative geotropism? Give one example of each type. | Geotropism: Growth response to gravity. Positive: Roots grow down. Negative: Shoots grow up. |
Geotropism is the growth movement of plants in response to gravity. Positive geotropism occurs when a plant part grows in the direction of gravitational pull, such as roots growing downward. Negative geotropism occurs when a plant part grows against gravitational pull, such as shoots growing upward. - Example of Positive Geotropism: Roots. - Example of Negative Geotropism: Shoots. |
| 36. (a) The variation of image distance (v) with object distance (u) for a convex lens is given in the observation table. Analyze it and answer the following questions: (i) Without calculation, find the focal length of the convex lens. Justify your answer. (ii) Which observation is not correct? Why? Draw a ray diagram to find the position of the image for this position of the object. (iii) Find the approximate magnification when u = −30 cm. |
(a) (i) Focal length ≈ 25 cm. (ii) Observation: u = −15 cm; v incorrect. (iii) Magnification: −5. |
(a) (i) When the object distance is equal to the focal length, the image distance is very large. Based on the table, focal length is approximately 25 cm. (ii) Observation with u = −15 cm shows a real image, but it should be virtual. The error is due to incorrect placement of the object. (iii) Magnification ( m = v/u = 150/−30 = −5 ). |
| OR 36.(b) (i) Define the principal axis of a lens. Draw a ray diagram showing the behavior of a light ray parallel to the principal axis of a concave lens. (ii) The focal length of a concave lens is 20 cm. Calculate the distance from the lens for placing a 5 cm tall object to form an image at 15 cm. Also, calculate the image size. |
(b) Parallel rays diverge. u = −60 cm; Image size = 1.25 cm. |
(b) (i) The principal axis is the line passing through the optical center and centers of curvature. In a concave lens, rays parallel to the principal axis appear to diverge from the focus. (ii) Using lens formula: ( 1/f = 1/v − 1/u ) ( f = −20 ),( v = −15 ):( u = −60 ) cm. Magnification:( m = v/u = −15/−60 = 1/4 ). Image size = ( 5 × 1/4 = 1.25 ) cm. |
| 37. Three metal samples (magnesium, aluminum, and iron) were treated with dilute HCl and HNO3. Observations for bubble formation and temperature were recorded. (a) In which test tube was the bubbling rate fastest? Why? (b) Which metal did not react with dilute HCl? Why? (c) (i) Why is hydrogen gas not evolved when a metal reacts with dilute HNO3? Name the ultimate products formed. |
(a) Magnesium. (b) Aluminum; oxide layer. (c) (i) Oxidizing agent; NO and H2O. |
(a) Bubbling was fastest in magnesium due to its high reactivity with acids, producing H2 gas. (b) Aluminum did not react with dilute HCl because its oxide layer (Al2O3) prevents reaction. (c) (i) HNO3 oxidizes H2 gas to form water. Products: Metal nitrate, NO, and H2O. |
| OR 37.(c) (ii) Name the reaction used to determine metal reactivity. How would you compare the reactivity of two metals X and Y? | (c) (ii) Displacement; compare reactions. | (c) (ii) Displacement reactions determine reactivity. Place metals in salt solutions of each other and observe displacement. The metal that displaces the other is more reactive. |
| 38. The kidneys are vital organs for survival. Describe the following: (a) Name the artery bringing oxygenated blood to the kidneys and the cluster of capillaries in the Bowman’s capsule. (b) Which organ stores urine? Is its activity controlled hormonally or nervously? (c) (i) List the two major steps involved in urine formation and their functions. |
(a) Renal artery; glomerulus. (b) Urinary bladder; nervous control. (c) (i) Ultrafiltration; selective reabsorption. |
(a) The renal artery delivers oxygenated blood to the kidneys. The glomerulus, a cluster of capillaries, filters blood in the Bowman’s capsule. (b) The urinary bladder stores urine and is under nervous control. (c) (i) Ultrafiltration: Blood pressure filters water, salts, and urea into the nephron. Selective reabsorption: Essential nutrients and water are reabsorbed into the bloodstream. |
| OR 38.(c) (ii) Where does selective reabsorption occur? What factors affect water reabsorption? | (c) (ii) PCT; ADH, hydration, salt levels. | (c) (ii) Selective reabsorption occurs in the PCT and DCT. Water reabsorption is affected by ADH levels, hydration state, and salt concentration in blood. |
| 39. Study the following circuit and answer: (a) Find the total resistance between points A and B. (b) Find the resistance between points B and C. (c) (i) Calculate the current drawn from the battery. |
(a) 26 Ω. (b) 4 Ω. (c) (i) 0.2 A. |
(a) Total resistance ( R_{AB} = 4 + 6 + 16 = 26 ) Ω. (b) Parallel resistance ( 1/R = 1/8 + 1/8 = 2/8 ), ( R_{BC} = 4 ) Ω. (c) (i) Total resistance: ( 26 + 4 = 30 ) Ω. Current: ( I = V/R = 6/30 = 0.2 ) A. |
| OR 39.(c) (ii) Which has more potential difference: 16 Ω resistor or the parallel combination of two 8 Ω resistors? Justify. | (c) (ii) 16 Ω; higher resistance. | (c) (ii) Potential difference ( V = IR ): 16 Ω resistor: ( V = 0.2 × 16 = 3.2 ) V. Parallel combination: ( V = 0.2 × 4 = 0.8 ) V. The 16 Ω resistor has a higher potential difference as it has greater resistance. |


































Comments