CBSE Class 10 Science Question Paper 2024 PDF (Set 3 - 31/4/3) is available for download here. CBSE conducted the Science exam on March 2, 2024, from 10:30 AM to 1:30 PM. The total marks for the theory paper are 80. The question paper contains 20% MCQ-based questions, 40% competency-based questions, and 40% short and long answer type questions.
CBSE Class 10 Science Question Paper 2024 (Set 3 - 31/4/3) with Answer Key
| CBSE Class 10 Science Question Paper 2024 (Set 3 - 31/4/3) with Answer Key | Check Solutions |
CBSE Science Question Paper (Set 3 – 31/4/3) 2024 Solution
| Question | Answer | Detailed Solution |
|---|---|---|
| 1. An electric iron of resistance 20Ω draws a current of 5A. The heat developed in the iron in 30 seconds is: (A) 15000 J (B) 6000 J (C) 1500 J (D) 3000 J |
(A) 15000 J | The heat developed in an electric circuit is calculated using Joule's Law of Heating: H = I2Rt. Given I=5A, R=20Ω, t=30s: H = (5)2 × 20 × 30 = 15000 J. |
| 2. The acid produced in our stomach during digestion and the base used to neutralise the excess acid during indigestion respectively are: (A) HCl, Mg(OH)2 (B) HCl, Ca(OH)2 (C) Amino acids, Ca(OH)2 (D) Lactic acid, Mg(OH)2 |
(A) HCl, Mg(OH)2 | HCl is the acid in our stomach aiding digestion. Mg(OH)2 (milk of magnesia) neutralises excess acid by reacting with it to form water and a salt. |
| 3. A zygote is formed by the fusion of a male gamete and a female gamete. The number of chromosomes in the zygote of a human is: (A) 23 (B) 44 (C) 46 (D) 92 |
(C) 46 | The zygote contains 46 chromosomes (23 pairs): 23 from the sperm and 23 from the egg, forming the diploid chromosome number in humans. |
| 4. The part of seed which is a source of food during germination of seed is: (A) Cotyledon (B) Radicle (C) Plumule (D) Embryo |
(A) Cotyledon | Cotyledons store food in the seed and provide energy during germination until the plant begins photosynthesis. |
| 5. Zn + 2CH3COOH → (CH3COO)2Zn + H2 The above reaction is a: (A) Decomposition reaction (B) Displacement reaction (C) Double displacement reaction (D) Combination reaction |
(B) Displacement reaction | Zinc displaces hydrogen from acetic acid to form zinc acetate and hydrogen gas, characteristic of a single displacement reaction. |
| 6. Four solutions, namely glucose, alcohol, hydrochloric acid, and sulphuric acid filled in four separate beakers are connected one by one in an electric circuit with a bulb. The solutions in which the bulb will glow when current is passed are: (A) Glucose and alcohol (B) Alcohol and hydrochloric acid (C) Glucose and sulphuric acid (D) Hydrochloric acid and sulphuric acid |
(D) Hydrochloric acid and sulphuric acid | Hydrochloric acid (HCl) and sulphuric acid (H2SO4) are strong electrolytes and dissociate into ions in water, enabling the bulb to glow. Glucose and alcohol are covalent compounds that do not produce ions. |
| 7. The metals which are found in both free state as well as combined state are: (A) Gold and platinum (B) Platinum and silver (C) Copper and silver (D) Gold and silver |
(C) Copper and silver | Copper and silver exist in free states due to low reactivity and in combined states as ores. Examples include native copper and silver sulphide (Ag2S). |
| 8. The part of the flower which attracts insects for pollination is/are: (A) Stigma and style (B) Sepals and petals (C) Petals only (D) Sepals only |
(C) Petals only | Petals are brightly colored and often fragrant, attracting insects for pollination. Sepals protect the flower bud and do not play a significant role in pollination. |
| 9. In human beings, when the process of digestion is completed, the (i) proteins, (ii) carbohydrates, and (iii) fats are respectively finally converted into: (A) (i) Amino acids, (ii) glucose, and (iii) fatty acids (B) (i) Amino acids, (ii) glucose, (iii) fatty acids and glycerol (C) (i) Glucose, (ii) fatty acids and glycerol, (iii) amino acids (D) (i) Sugars, (ii) amino acids, (iii) fatty acids and glycerol |
(B) (i) Amino acids, (ii) glucose, (iii) fatty acids and glycerol | Proteins break into amino acids, carbohydrates into glucose, and fats into fatty acids and glycerol during digestion. These products are absorbed and utilized for energy and growth. |
| 10. The number of single and double bonds present in a molecule of benzene (C6H6) respectively, are: (A) 6 and 6 (B) 9 and 3 (C) 3 and 9 (D) 3 and 3 |
(B) 9 and 3 | Benzene has 9 single bonds (6 C-H and 3 C-C) and 3 double bonds (C=C). The alternating double bonds give benzene its resonance stability. |
| 11. A plant growth inhibitor hormone which causes wilting of leaves is called: (A) Auxin (B) Cytokinin (C) Abscisic acid (D) Gibberellin |
(C) Abscisic acid | Abscisic acid is a stress hormone that induces wilting by closing stomata, reducing water loss during drought conditions. |
| 12. Some wastes are given below: (i) Garden waste (ii) Ball point pen refills (iii) Empty medicine bottles made of glass (iv) Peels of fruits and vegetables (v) Old cotton shirt The non-biodegradable wastes among these are: (A) (i) and (ii) (B) (ii) and (iii) (C) (i), (iv) and (v) (D) (i), (iii) and (iv) |
(B) (ii) and (iii) | Non-biodegradable wastes like ballpoint pen refills (plastic) and glass bottles do not decompose naturally, unlike garden waste, fruit peels, and cotton shirts. |
| 13. A rectangular loop carrying a current I is situated near a straight conductor carrying current. The straight conductor will: (A) Remain stationary (B) Move towards the loop (C) Move away from the loop (D) Rotate about its axis |
(B) Move towards the loop | Parallel currents attract each other due to the magnetic fields produced. The straight conductor moves towards the loop. |
| 14. Absolute refractive index of glass and water is 3/2 and 4/3 respectively. If the speed of light in glass is 2 × 108 m/s, the speed of light in water is: (A) 9/4 × 108 m/s (B) 5/2 × 108 m/s (C) 7/3 × 108 m/s (D) 16/9 × 108 m/s |
(A) 9/4 × 108 m/s | Using the formula n = c/v, where c is the speed of light in vacuum, calculate speed in water: v = c × (4/3). |
| 15. When a beam of white light passes through a region having very fine dust particles, the colour of light mainly scattered is: (A) Red (B) Orange (C) Blue (D) Yellow |
(C) Blue | Blue light, with a shorter wavelength, is scattered more than other colors due to Rayleigh scattering, making the region appear blue. |
| 16. Consider the following combinations of resistors: The combinations having equivalent resistance 1Ω is/are: (A) I and IV (B) Only IV (C) I and II (D) I, II and III |
(C) I and II | For I and II, calculate equivalent resistances using series and parallel rules. Both configurations lead to an equivalent resistance of 1Ω. |
| 17. Assertion (A): Oxygen is essential for all aerobic forms of life. Reason (R): Free oxygen atoms combine with molecular oxygen to form ozone. (A) Both A and R are true, and R is the correct explanation of A. (B) Both A and R are true, but R is not the correct explanation of A. (C) A is true, but R is false. (D) A is false, but R is true. |
(B) Both A and R are true, but R is not the correct explanation of A. | Oxygen is required for aerobic respiration. Though ozone forms through the combination of oxygen atoms and molecules, it is not related to oxygen's necessity for aerobic life. |
| 18. Assertion (A): Magnetic field lines never intersect each other. Reason (R): If they intersect, then at the point of intersection, the compass needle would point towards two directions, which is not possible. (A) Both A and R are true, and R is the correct explanation of A. (B) Both A and R are true, but R is not the correct explanation of A. (C) A is true, but R is false. (D) A is false, but R is true. |
(A) Both A and R are true, and R is the correct explanation of A. | Magnetic field lines represent the direction of the magnetic field at a point. They cannot intersect because this would imply two directions for the magnetic field at the same point, which is impossible. |
| 19. Assertion (A): The extraction of metals from their sulphide ores cannot take place without roasting of the ore. Reason (R): Roasting converts sulphide ores directly into metals. (A) Both A and R are true, and R is the correct explanation of A. (B) Both A and R are true, but R is not the correct explanation of A. (C) A is true, but R is false. (D) A is false, but R is true. |
(B) Both A and R are true, but R is not the correct explanation of A. | Roasting converts sulphide ores into oxides, which are then reduced to metals. It does not directly convert sulphide ores into metals. |
| 20. Assertion (A): In the human heart, ventricles have thicker muscular walls than atria. Reason (R): Ventricles have to pump the blood into various organs. (A) Both A and R are true, and R is the correct explanation of A. (B) Both A and R are true, but R is not the correct explanation of A. (C) A is true, but R is false. (D) A is false, but R is true. |
(A) Both A and R are true, and R is the correct explanation of A. | Ventricles pump blood over longer distances and require greater pressure, which is why their muscular walls are thicker. |
| 21. (a) We need to water the soil in plants on a regular basis. But it ultimately reaches the leaves of the plant. Explain how this takes place. | The ascent of sap | Water reaches the leaves through the ascent of sap, primarily driven by transpiration pull. Water is absorbed by roots via osmosis, moves across the root cortex to the xylem, and is pulled upward due to cohesion, adhesion, and transpiration at the leaves. |
| OR 21. (b) Name the type of nutrition exhibited by Amoeba. Explain how food is taken in and digested by this organism. | Holozoic Nutrition | Amoeba exhibits holozoic nutrition. It engulfs food particles with pseudopodia to form a food vacuole. Enzymes are secreted into the vacuole to digest the food, and nutrients are absorbed into the cytoplasm. Undigested matter is egested out of the cell. |
| 22. When a soap is dissolved in water, the soap molecules form structures. What are these structures called? Draw a labelled diagram of these structures. | Micelles | Soap molecules form micelles with hydrophobic tails clustering inward and hydrophilic heads outward. This arrangement traps grease and dirt, allowing it to be washed away. |
| 23. (a) 1 gram of solid sodium chloride was taken in a clean and dry test tube and concentrated sulphuric acid was added to it. (i) Name the gas evolved in the reaction. (ii) What will be observed when this gas is tested with (I) dry, and (II) wet blue litmus paper? Write your conclusion about the nature (acidic/basic) of this gas. |
(i) Hydrogen Chloride (HCl) (ii) Acidic in nature |
(i) The reaction produces HCl gas. (ii) Dry blue litmus shows no change, but wet blue litmus turns red because HCl dissolves in water to form hydrochloric acid. This indicates the gas is acidic in nature. |
| OR 23. (b) Some metals react with acids to produce salt and hydrogen gas. Illustrate it with an example. How will you test the presence of this gas? | Zn + H2SO4 → ZnSO4 + H2 | When zinc reacts with dilute sulfuric acid, it forms zinc sulfate and hydrogen gas. The presence of hydrogen is tested using the “pop” test: bringing a burning splinter near the test tube produces a popping sound if hydrogen is present. |
| 24. Explain how equal genetic contribution of male and female parents is ensured in the progeny. | By Meiosis and Fertilization | Each parent contributes 23 chromosomes through their gametes (haploid cells). During fertilization, these combine to restore the diploid chromosome number (46) in the zygote. |
| 25. What would have been the colour of the sky, if the Earth had no atmosphere? Give reason to justify your answer. | Black | Without an atmosphere, there would be no scattering of sunlight. As a result, the sky would appear black, similar to outer space, where sunlight travels without being scattered. |
| 26. Calculate the resistance of a copper wire of length 1000 m and area of cross-section 2 mm2. Resistivity of copper is 1.6 × 10-8 Ωm. | 8 Ω | Resistance R is given by the formula: R = ρ(L/A), where: - ρ = 1.6 × 10-8 Ωm - L = 1000 m - A = 2 mm2 = 2 × 10-6 m2 Substitute values: R = (1.6 × 10-8 × 1000) / (2 × 10-6) = 8 Ω. |
| 27. Define the term power of accommodation of human eye. What happens to the image distance in the eye when we increase the distance of an object from the eye? Name and explain the role of the part of human eye responsible for it in this case. | Lens and Ciliary Muscles | The power of accommodation is the ability of the eye to adjust the focal length of its lens to focus on objects at varying distances. When the object distance increases, the image distance remains fixed (on the retina). The ciliary muscles relax, making the lens thinner and increasing its focal length to focus the object on the retina. |
| 28. Write chemical equations for the following reactions, giving the conditions for the reaction in each case: (a) Reaction of ethanol with ethanoic acid (b) Reaction of an ester with a base (NaOH) (c) Formation of ethene from ethanol |
Chemical Reactions | (a) C2H5OH + CH3COOH → CH3COOC2H5 + H2O (in presence of conc. H2SO4) (b) CH3COOC2H5 + NaOH → CH3COONa + C2H5OH (c) C2H5OH → C2H4 + H2O (with conc. H2SO4, 443K). |
| 29. A student fixes a sheet of white paper on a drawing board. He places a bar magnet in the center and sprinkles iron filings. The filings arrange in a specific pattern. Answer the following: (a) Why do iron filings arrange in a particular pattern? (b) What does the crowding of iron filings at the ends of the magnet indicate? (c) What do the lines, along which the iron filings align, represent? |
Magnetic Field | (a) Iron filings align due to the magnetic field produced by the bar magnet. (b) Crowding of filings at the poles indicates stronger magnetic fields near the poles. (c) The lines represent magnetic field lines. |
| 30. (a) Name any two pairs of contrasting characters other than height used by Mendel in his experiments. | Flower color and Seed shape | Examples of contrasting characters: (1) Flower color: Purple and White. (2) Seed shape: Round and Wrinkled. Mendel used these traits to establish laws of inheritance. |
| (b) On crossing a tall and a short pea plant, Mendel obtained F1 generation in which all pea plants were tall. (i) Are these plants exactly the same as tall plants of the parent generation? Write their gene combination. (ii) Give the percentage of short plants obtained in F2 generation when F1 plants are self-pollinated. (iii) In what ratio would you find TT and Tt in F2 generation? |
(i) No, Tt (ii) 25% (iii) 1:2 |
(i) The F1 generation plants are heterozygous tall (Tt), whereas the tall parent plant was homozygous dominant (TT). (ii) When F1 plants are self-pollinated, the genotypic ratio in F2 generation is 1:2:1 (TT:Tt:tt). Short plants (tt) make up 25%. (iii) Among the tall plants, TT:Tt is in the ratio 1:2. |
| 31. Write the common name and the chemical name of the compound CaSO4 · ½ H2O. Write the method of its preparation. Give chemical equation for the reaction, when water reacts with CaSO4 · ½ H2O. | Common Name: Plaster of Paris (POP) Chemical Name: Calcium Sulphate Hemihydrate |
Plaster of Paris is prepared by heating gypsum (CaSO4 · 2H2O) to 373 K: CaSO4 · 2H2O → CaSO4 · ½ H2O + 1½ H2O. Reaction with water: CaSO4 · ½ H2O + 1½ H2O → CaSO4 · 2H2O (exothermic reaction forming gypsum). |
| 32. (a) Explain with the help of a labelled diagram, the process of reproduction in Hydra by budding. Name the cells used for reproduction in this process. | Reproductive cells; Budding | Hydra reproduces asexually by budding. A bud grows on the parent body, matures, and detaches to form a new Hydra. Diagram: Shows parent Hydra, growing bud, and separated offspring. |
| (b) List two roles of each of the following in the human reproductive system: (i) Seminal vesicles and prostate gland (ii) Oviduct (iii) Testis |
See explanation | (i) Seminal Vesicles and Prostate Gland: - Seminal vesicles secrete fluid rich in fructose to nourish sperm. - Prostate gland secretes an alkaline fluid to neutralize vaginal acidity. (ii) Oviduct: - Site of fertilization between egg and sperm. - Transports the zygote to the uterus. (iii) Testis: - Produces sperm cells via spermatogenesis. - Secretes testosterone, responsible for male secondary sexual characteristics. |
| 33. Differentiate between biodegradable and non-biodegradable wastes. Write two harmful effects of non-biodegradable wastes if not disposed of properly. | Biodegradable decomposes; Non-biodegradable persists | Biodegradable: Decomposes naturally (e.g., fruit peels). Non-biodegradable: Persists long-term (e.g., plastics). Harmful effects: (1) Environmental pollution due to accumulation. (2) Health hazards from toxic releases like dioxins. |
| 34. (a) Define a reflex arc. Why have reflex arcs evolved in animals? Trace the sequence of events when touching a hot object. | Reflex arc ensures rapid response | A reflex arc is a neural pathway for automatic response to stimuli. It evolved for quick protection from harm. Sequence: Stimulus → Sensory neuron → Spinal cord → Motor neuron → Effector (muscle contracts). |
| (b) (i) Leaves of ‘chhui-mui’ plant begin to fold up and droop in response to a stimulus. Name the stimulus and write the cause for such a rapid movement. Is there any growth involved in the movement? | Touch; No growth involved | The stimulus is touch (mechanical stimulation). Rapid movement occurs due to a sudden change in turgor pressure in specialized cells (pulvini). When stimulated, water moves out of these cells, causing them to lose rigidity and resulting in drooping leaves. This is a nastic movement, which is independent of growth. |
| (ii) Define geotropism in plants. What is meant by positive and negative geotropism? Give one example of each type. | Positive: Roots; Negative: Shoots | Geotropism is the directional growth of plants in response to gravity. Positive geotropism: Growth towards gravity (e.g., roots growing into soil). Negative geotropism: Growth away from gravity (e.g., shoots growing upward towards sunlight). |
| 35.(a) What is a chemical reaction? Describe one activity each showing (i) change of colour, (ii) change in temperature. | (i) Fe + CuSO4 (ii) CaO + H2O |
A chemical reaction involves the rearrangement of atoms. (i) Fe + CuSO4 → FeSO4 + Cu (blue fades, brown deposit). (ii) CaO + H2O → Ca(OH)2 (exothermic reaction produces heat). |
| (b) (i) Define a decomposition reaction. How can we say that (I) electrolysis of water, and (II) blackening of silver bromide when exposed to sunlight, are decomposition reactions? Mention the type of energy involved in each case. | Breakdown of compounds; Electrical and Light energy | A decomposition reaction is one where a single compound breaks into two or more simpler substances. (I) Electrolysis of water: H2O → H2 + O2 - Decomposition due to electrical energy. (II) Blackening of silver bromide: 2AgBr → 2Ag + Br2 - Decomposition due to light energy. |
| (ii) The type of reactions in which (I) calcium oxide is formed, and (II) calcium hydroxide is formed are opposite reactions to each other. Justify this statement with chemical equations. | Decomposition and Combination | (I) Formation of calcium oxide: CaCO3 → CaO + CO2 (Decomposition). (II) Formation of calcium hydroxide: CaO + H2O → Ca(OH)2 (Combination). These are opposite as one breaks down a compound while the other combines substances. |
| 36. (a) (i) State the laws of reflection of light. | Two laws of reflection | (1) The angle of incidence (∠i) is equal to the angle of reflection (∠r). (2) The incident ray, the reflected ray, and the normal to the surface at the point of incidence all lie in the same plane. |
| (ii) An object of height 5.0 cm is placed at 15 cm in front of a concave mirror of focal length 10 cm. At what distance from the mirror should a screen be placed, so that a focused image is obtained on it? Find the height of the image. | Image distance = 30 cm Image height = -10 cm |
Using the mirror formula: 1/f = 1/v + 1/u Given: f = -10 cm, u = -15 cm. Calculation: 1/v = 1/f - 1/u = -1/10 - (-1/15) = -3/30 + 2/30 = -1/30. v = -30 cm (negative sign indicates real image). Magnification (m) = -v/u = -(-30)/(-15) = -2. Image height (h') = m × object height (h) = -2 × 5 = -10 cm (inverted image). Screen should be placed 30 cm in front of the mirror. |
| OR 36.(b) (i) State the laws of refraction of light. | Two laws of refraction | (1) The incident ray, the refracted ray, and the normal to the surface at the point of incidence all lie in the same plane. (2) The ratio of the sine of the angle of incidence to the sine of the angle of refraction is constant (Snell’s Law): sin(i)/sin(r) = n (refractive index). |
| (ii) Draw a ray diagram to show refraction of a ray of light through a rectangular glass slab. How is the emergent ray related to the incident ray? Mark lateral displacement in the diagram. | Emergent ray is parallel to incident ray | The emergent ray is parallel to the incident ray but is laterally displaced due to refraction. Diagram: - Incident ray enters the glass slab and bends towards the normal (denser medium). - Inside the slab, it travels in a straight line. - Emergent ray exits the slab, bending away from the normal and appearing parallel to the incident ray. |
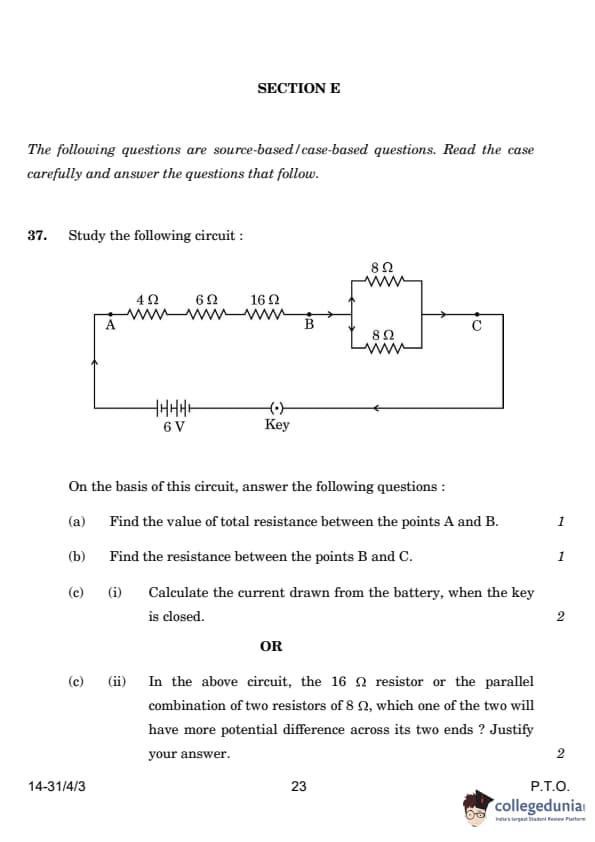
| 37. Study the following circuit: (a) Find the total resistance between points A and B. (b) Find the resistance between points B and C. (c) Calculate the current drawn from the battery when the key is closed. |
(a) 26 Ω (b) 4 Ω (c) 0.2 A |
(a) Total resistance between A and B: RAB = 4 Ω + 6 Ω + 16 Ω = 26 Ω (series combination). (b) Resistance between B and C: 1/RBC = 1/8 + 1/8 = 2/8 = 1/4. RBC = 4 Ω (parallel combination). (c) Total resistance in the circuit: Rtotal = RAB + RBC = 26 Ω + 4 Ω = 30 Ω. Current (I) = V/R = 6 V / 30 Ω = 0.2 A. |
| 37.c.(ii) Compare the potential difference across the 16 Ω resistor and the parallel combination of two 8 Ω resistors. | Potential difference across 16 Ω is higher | The 16 Ω resistor will have a higher potential difference as it has a higher resistance compared to the parallel combination of the two 8 Ω resistors. V = IR applies. |
| 38. Three metal samples (magnesium, aluminum, and iron) were reacted with dilute hydrochloric acid, and the following questions were asked: (a) In which test tube will the rate of bubble formation be the fastest, and why? (b) Which metal does not react with dilute HCl? Give a reason. (c) (i) Why is hydrogen gas not evolved when metals react with dilute nitric acid? |
(a) Magnesium (b) Copper (c) (i) Nitric acid oxidizes hydrogen |
(a) Magnesium reacts fastest because it is the most reactive among magnesium, aluminum, and iron. It reacts vigorously with HCl, producing hydrogen gas bubbles rapidly. Reaction is highly exothermic. (b) Copper does not react with dilute HCl as it is less reactive than hydrogen. Copper cannot displace hydrogen from HCl to produce hydrogen gas. (c) (i) Hydrogen gas is not evolved when metals react with dilute nitric acid because nitric acid is a strong oxidizing agent. It oxidizes the hydrogen gas formed during the reaction into water. |
| 39. c.(ii) Write the ultimate products formed in such a reaction. | (ii) Metal nitrate and water |
(ii) The reaction forms metal nitrate and water as products. Example: Zn + 2HNO3 → Zn(NO3)2 + H2O |
| 39.Kidneys are vital organs for survival. In case of kidney failure, an artificial kidney is used. (a) (i) Name the artery that brings oxygenated blood to the kidney. (ii) Name the cluster of capillaries in the Bowman’s capsule. (b) Name the organ that stores urine. Is it under hormonal or nervous control? (c) (i) List two major steps in urine formation. |
(a) (i) Renal artery (ii) Glomerulus (b) Urinary bladder; Nervous control (c) (i) Filtration and reabsorption |
(a) (i) The renal artery supplies oxygenated blood to the kidneys. (ii) The glomerulus is a cluster of capillaries involved in filtration in the Bowman’s capsule. (b) The urinary bladder stores urine and is under nervous control for voluntary and involuntary urination. (c) (i) Urine formation involves: - Filtration: Removes waste and water from the blood in the glomerulus. - Reabsorption: Essential nutrients and water are absorbed back in the renal tubules. |
| OR 39.(ii) In which part of the nephron does selective reabsorption occur? Name two factors that influence water reabsorption. | (ii) Proximal convoluted tubule; ADH and hydration level | (ii) Selective reabsorption occurs in the proximal convoluted tubule. Water reabsorption depends on: - Hormonal control (ADH). - Hydration level and solute concentration in filtrate. |

































Comments