CBSE Class 10 Science Question Paper 2026 Set-2 (Code: 31/5/2) is now available for download. CBSE conducted the Class 10 Science examination on Feb 25, 2026, from 10:30 AM to 1:30 PM. The question paper consists of 35 questions carrying a total of 80 marks. Part A is compulsory for all candidates. Part B has two options. Candidates have to attempt only one of the given options. Option I: Physics and Option II: Chemistry. The Science question paper 2026 was rated moderately easy by the students.
CBSE Class 10 Science Question Paper 2026 (Set 2- 31/5/2) with Answer Key
Candidates can use the link below to download the CBSE Class 10 Science 2026 Set 2 Question Paper with detailed solutions.
| CBSE Class 10 2026 Science Question Paper Set 2 with Solutions | Download PDF | Check Solution |

In human beings, the implantation of fertilised egg takes place in which part of female reproductive system?
Yeast multiplies by which of the following methods?
Identify the part that controls the closing and opening of the stomatal pore in leaves of plants. Select the correct option.
Choose the equation of reaction that correctly represents anaerobic respiration in muscles:
Which of the following is a non-biodegradable pollutant?
When a human egg is fertilized by a sperm having ‘Y’ chromosome, the zygote has the following combination of chromosomes:
The reasons for excessive generation of wastes are:
(i) Use and throw policy.
(ii) Increased availability of packaged food.
(iii) Increased construction wastes.
(iv) Non-sorting of dry and wet wastes.
Options:
Assertion (A): The peristaltic movements occur all along the gut.
Reason (R): The lining of the gut has muscles that contract rhythmically to push the food in regulated manner.
Assertion (A): Bacteria produced as a result of asexual reproduction would be similar.
Reason (R): There would be only minor differences between the bacteria generated due to small inaccuracies in DNA replication.
Observe the given figure and identify the labelled parts P, Q, R and S.

Which of the plant hormones are responsible for the following processes?
(i) Promote cell division
(ii) Inhibition of growth
(iii) Detection of light
(iv) Wilting of leaves
Briefly mention the steps in double-circulation through human heart.
Given below is a diagrammatic representation of cross-section of a leaf:
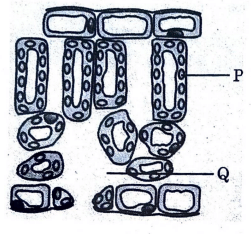
(i) Identify ‘P’ in the given diagram and write down its role in plants.
(ii) Write down a balanced equation of process of photo-synthesis.
Mention any one harmful effect of using plastic bags on the environment. Suggest better alternatives to the usage of plastic bags.
Paddy fields require a large amount of water. The pesticides and chemical fertilizers used are washed down into the soil or waterbodies. How do these chemicals reach our bodies? What is this phenomenon known as?
Mention the gland and the hormone secreted by it in scary situation in human beings.
Write two responses that enable the human body to be ready to deal with such situation.
Write the structure and function of Bowman’s capsule.
Write the structure and function of Bowman’s capsule.
What is excretion? Why is it necessary for any living organism?
State two similarities between lungs and kidneys.
(i) Sugarcane does not produce seeds so name the process through which it will be able to reproduce.
(ii) List any two advantages of this method.
(iii) Besides sugarcane, give two more examples of plants that reproduce by this method.
(iv) Why regeneration is not possible in all the types of animals?
(i) State the changes taking place in the following structures of flower post fertilization:
(a) Zygote
(b) Ovule
(c) Ovary
(d) Sepals
(ii) Define germination.
Which of the following set of compounds does not belong to same homologous series?
Which of the following is an amphoteric oxide?
In the following diagram, if acetic acid of same concentration is taken in place of dilute sulphuric acid, then
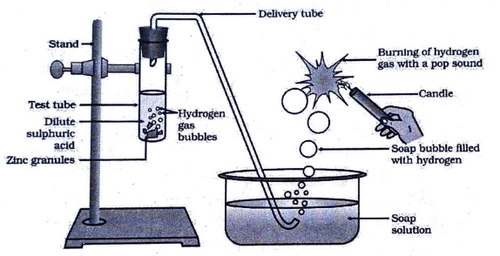
The volume ratio of hydrogen and oxygen gases liberated during electrolysis of water is:
Study the following table and select the correct option:
Which of the following will not undergo addition reaction?
On reaction with dilute HCl, which of the following pair of metals will evolve hydrogen gas?
Assertion (A): Reaction of quick lime with water is an exothermic reaction.
Reason (R): A large amount of heat is evolved on the reaction of quick lime and water.
How is a universal indicator obtained? How is the wide range of pH of solution tested by it?
What happens when
(i) Calcium carbonate is heated?
(ii) Silver bromide is exposed to sunlight?
(iii) Lead is added to copper (II) chloride solution?
Write balanced chemical equations of the reactions involved in support of your answer.
(a) Give the chemical name and formula of Plaster of Paris.
(b) Write the chemical equation of its preparation.
(c) Give any two uses of it.
(a) Name the acid present in ant’s sting.
(i) While diluting an acid, it is recommended that the acid should be added to water.
(ii) Baking soda is used as an antacid.
Write structural formula and name of 4\(^{th}\) member of alcohol homologous series.
(b) What happens when ethanol is heated with alkaline KMnO\(_4\)? Write chemical equation involved.
(c) Write the chemical equation of reaction of ethanol with ethanoic acid in the presence of concentrated H\(_2\)SO\(_4\). Write the name of this reaction.
(c) What happens when ethanol is heated with excess concentrated sulphuric acid at 443 K? Write chemical equation involved. What is the role of concentrated sulphuric acid in this reaction?
(i) Give reasons for the following:
(I) Ionic compounds have generally high melting points and boiling points.
(i) Give reasons for the following:
(I) Ionic compounds have generally high melting points and boiling points.
(II) Solder, an alloy of lead and tin, is used for welding electrical wires.
(III) Carbon cannot reduce the oxides of Na or Mg.
(III) Carbon cannot reduce the oxides of Na or Mg.
(ii) Give reasons for the following:
(I) The wires carrying current in homes have a coating of PVC.
(iii) Show the formation of ionic compound CaO with electron dot structure.
To restore clear vision in persons whose size of the eyeball has reduced, he/she is suggested to use suitable
Rays from the sun converge at a point 25 cm behind a convex lens. The distance at which an object be placed in front of the lens to get a virtual image, is:
Assertion (A): The needle of a magnetic compass kept in strong external magnetic field, always aligns itself in north-south direction on the earth.
Reason (R): Behaviour of the needle of a compass is same as that of a freely suspended bar magnet.
(a) Define absolute refractive index of an optical medium.
(b) Arrange the given material mediums in increasing order of speed of light through them.
Given Refractive Indices:
A = 1.50
B = 1.46
C = 1.31
D = 1.77
(i) How does the change in curvature of the eye lens help us in the process of seeing the nearby objects clearly?
(ii) State the range of the power of accommodation of a normal human eye.
Draw a ray diagram to show the correction of eye defect of an old man who cannot see an object placed closer than 1 m from his eye clearly.
Why does an electric bulb become dim when an electric heater in parallel circuit is switched ON?
(b) How to connect three resistors each of resistance 8 \(\Omega\), so that the equivalent resistance of the combination is 12 \(\Omega\)? Draw diagram of the combination and justify your answer.
An object is placed at a distance of 30 cm in front of a convex lens of focal length 15 cm. Use lens formula to determine the position of the image. What will be the size of the image in this case?
(a) Draw the ray diagram for refraction of light through a glass prism and mark angle of refraction and angle of deviation.
(b) When the path of a light ray refracted through a glass prism is reversed, how will the angle of deviation change? Explain.
Passage:
Three students Shweta, Ayesha and Samridhi were performing i an
experiment to understand the factors on which the resistance of a conductor depends. Each one of them completed electric circuit with the help of a cell, an ammeter, a plug key and wire.
Shweta put nichrome wire of length 7’ in the circuit and after plugging the key, noted current in the ammeter.
Ayesha put nichrome wire of same thickness but twice the length i.e. ‘21’ in the circuit and after plugging the key, noted current in the ammeter.
Samridhi took copper wire of length ‘I’ and same thickness in the circuit and after plugging the key, noted current in the ammeter.
(a) If the ammeter reading is X ampere with nichrome wire of length \(l\), then what will be the ammeter reading if the length of nichrome wire is doubled with same area of cross-section?
(b) What happens to the ammeter reading if the area of cross-section of nichrome wire is doubled, keeping the length of wire \(l\) the same?
(c) Define resistivity. Write its SI unit. Compare the resistivity of an alloy with its constituent metals.
(i) Tungsten is used almost exclusively for making the filament of electric lamps.
(i) Tungsten is used almost exclusively for making the filament of electric lamps.
(i) List any two features of the electric current used in the houses of India.
(ii) Write any two differences between direct current and alternating current.
(iii) How will you identify live wire and neutral wire in a domestic electric circuit?
(ii) Distinguish between overloading and short-circuiting of a circuit.
(iii) Giving reason explain what type of materials are used in fuse wires.





































Comments