
Education Journalist | Study Abroad Strategy Lead
Hafnium is a chemical element with the atomic number 72 and the symbol Hf. It is a tetravalent transition metal that is silvery grey and glossy. This element, which is present in some zirconium minerals, shares chemical similarities with zirconium. Both electrodes and filaments employ hafnium. Hafnium is sometimes combined with other elements like titanium, tungsten, or niobium in some superalloys used for particular purposes.
| Table of Content |
Key Terms: Hafnium, Atomic Number, Transition Metal, Zirconium, Electrons, Modern Periodic Table, Corrosion
Hafnium
[Click Here for Sample Questions]
Hafnium can be classified as a tetravalent transition metal with atomic number 72. It is represented with the symbol Hf in the Modern Periodic Table. The element Hf was named after the city in which it was discovered, Copenhagen, whose Latin name is Hafnia.

Hafnium
Also Read:
Occurrence of Hafnium
[Click Here for Sample Questions]
The upper crust of Earth contains nearly 5.8 ppm of hafnium. Earth does not retain it as a free element. As an alternative, this metal can be obtained by combining a solid solution with natural zirconium compounds, such as ZrSiO4, and zircon, which usually replace about 1-4% of the Zr by Hf.
Rarely, hafnon, with atomic Hf greater than Zr, is formed during crystallization when the Hf/Zr ratio increases. Zircon varieties with unusually high Hf contents are known as alvite.
Also Read: Classification of Crystalline Solids
Physical Properties of Hafnium
[Click Here for Sample Questions]
The physical properties of Hafnium are as follows:
- Hafnium is a bright, silvery metal with ductile properties.
- It often demonstrates corrosion resistance and has many chemical characteristics with zirconium. This resemblance is caused by the existence of identity and belonging to the same group of valence electrons.
- The hafnium metal is significantly impacted by the zirconium impurities. As a result, they are both regarded as being the most inseparable.
- A thin film of a protective coating is created on the metal when it reacts with water, indicating the presence of corrosion.
- Acids have no impact on the metal, although it still emits oxides when burnt in the presence of oxygen. To some of the concentrated alkalis, this metal is quite resilient.
- This metal, which makes up around 5.8 ppm of the top crust of the planet, is not present naturally in the environment.
Chemical Properties of Hafnium
[Click Here for Sample Questions]
The chemical properties of Hafnium are listed below:
- In the presence of air, hafnium reacts to create a shielding coating that stops further corrosion. Although this element is not easily harmed by acids, it may be burned in the air or oxidised with halogens. Finely split hafnium may spontaneously ignite in the air, just like its sibling metal zirconium, making it very resistant to concentrated alkali.
- Zirconium and hafnium have chemistry that prevents their separation through different chemical processes.
- The main variations in these twin elements' chemistry are their solubility in solvents and the melting and boiling temperatures of the compounds.
Read More: Ruthenium
Applications of Hafnium
[Click Here for Sample Questions]
The major applications of Hafnium are as follows:
- Hafnium has the unusual ability to absorb neutrons. As a result, it is utilised in nuclear submarine control rods as well as reactors.
- Due to their higher melting points, its compounds are employed in the ceramic and alloy industries.
- It is frequently used in microprocessors because they help to reduce leakage current.
- Additionally, it is utilised in gas-filled and incandescent lights.
- This metal emits electrons, which act as an electrode during the plasma cutting process.
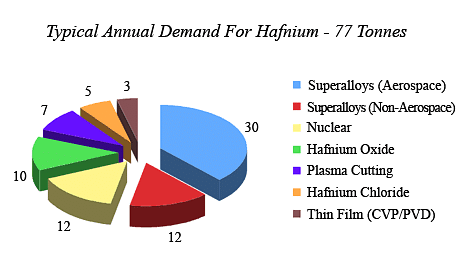
Applications of Hafnium
Production of Hafnium
[Click Here for Sample Questions]
Most of the zirconium that is extracted comes from the heavy mineral sands or deposits of rutile and ilmenite, which also provide most of the hafnium. Zirconium is one of the best metals for nuclear fuel rod cladding as it has strong chemical stability, as well as a very low neutron capture cross-section at high temperatures.
Zirconium impurities often make it significantly less suitable for any usage in nuclear reactors due to hafnium's neutron-absorbing characteristics. As a result, for hafnium and zirconium to be used in nuclear power, a full separation is needed. The main source of hafnium is the manufacturing of zirconium without hafnium.
Precautions
[Click Here for Sample Questions]
Hafnium metal should be handled carefully when machined since it is pyrophoric and the small particles can spontaneously ignite when exposed to air. Hafnium-containing compounds are not commonly found.
It is not thought to be hazardous to use this pure metal. Hafnium compounds, however, need to be treated cautiously as if they were hazardous. Ionic forms of the metal are often more poisonous, and only a small amount of animal research has been done on these substances. Hafnium exposure can occur at work through inhalation, ingestion, eye and skin contact, and breathing.
Read More: Rutherfordium
Things to Remember
- Hafnium is a tetravalent transition metal which is silver-grey and glossy in appearance.
- The element has the atomic number 72 and the symbol Hf.
- Hafnium exhibits corrosion resistance and has various similar chemical characteristics to zirconium.
- Owing to its ability to absorb neutrons, Hafnium is used in nuclear submarine control rods as well as reactors.
- Hafnium is also utilized in gas-filled and incandescent lights.
Sample Questions
Ques. List down the properties of Hafnium. (3 Marks)
Ans. Hafnium is classified as a shiny and silvery metal that shows ductile features. This element is usually resistant to corrosion and shows similar chemical properties to that of zirconium. There are similarities between Hafnium and Zirconium because of the presence of valence electrons which are the same and that of the same group.
Ques. What is a transition metal? (3 Marks)
Ans. The elements that partly fill the ‘d’ and ‘f’ subshells are termed Transition Elements or simply Transition Metals. In general, IUPAC clearly elaborates that in a transition element ‘d’ shells partly are covered with electrons. Transition elements also possess the capability to form certain stable cations that have incomplete ‘d’ shells.
Ques. Mention the reasons why transition elements form interstitial compounds. (3 Marks)
Ans. An interstitial compound, or simply an interstitial alloy, is a compound that forms when a small atom lays the foundation in an interstitial hole in a metal lattice. There are several interstitial compounds formed with help of transition metals since the vacant spaces of the transition elements are seen to become filled by small atoms. Transition metals are renowned for forming interstitial compounds since they have a closed structure with several voids present in them.
Ques. Describe the chemical activities of Hafnium. (3 Marks)
Ans. Hafnium reacts with air to create a protective coating that helps in stopping further corrosion. Though hafnium is not easily harmed by acids, it may burn in the air or oxidise with halogens. Just like its sibling element zirconium, finely split hafnium can also spontaneously ignite in the air, making it very resistant to concentrated alkali.
Ques. List down a few metallic properties of transition metals. (3 Marks)
Ans. These elements have various metallic properties such as ductility, malleability, high tensile strength, metallic lustre and more. They are commonly good conductors of heat and electricity. The reason why they exhibit metallic qualities is that they come with low ionization energies, as well as numerous empty orbitals in their outer shells.
Ques. Transition metals generally form coloured compounds. Why? (3 Marks)
Ans. Transition elements are very reactive, and they form coloured compounds. Transition elements have unpaired electrons. These unpaired electrons undergo d-d transition by absorption of energy, and they have a visible region, and then they emit light and show complementary colours. This is the reason why transition elements form coloured compounds.
Ques. What are the dangers related to Hafnium? (3 Marks)
Ans. As such Hafnium has no known toxicity. Hafnium is completely insoluble in water, saline solutions or body chemicals. One can be exposed to Hafnium through inhalation, ingestion, and eye or skin contact. If overexposed to hafnium and its compounds, one may see slight irritation of the eyes, skin, and mucous membranes.
Ques. Is hafnium element rare or common? (3 Marks)
Ans. Hafnium is the 45th most abundant element on the surface of the earth. According to Chemicool, hafnium comprises about 3.3 parts per million (ppm) of the Earth's crust by weight. The element is quite resistant to corrosion because of the formation of an oxide film on exposed surfaces.
Ques. Why are transition metals also termed ‘noble metals’? (2 Marks)
Ans. The reason behind this is that these have low enthalpies of hydration and high enthalpy of ionization. As a result, these metals are highly resistant to acids. However, these metals react to acidic mixtures like aqua regia, which is a mixture of hydrochloric acid and nitric acid. But silver doesn’t dissolve in aqua regia too.
Ques. What are inner transition elements? (4 Marks)
Ans. The inner transition elements are f-block elements. Their 4f and 5f orbitals are filled and their series are known as lanthanoids. Their third last shell is filled with electrons. These transition elements form coloured ions and show varied valences. They are radioactive and synthetic beyond atomic number 92 up to 103. They are not found in nature in the earth’s crust.
The inner transition elements are found in two series namely the 4f series known as lanthanoids and the 5f series known as actinoids. The Th, U and Pa provide nuclear energy. Their outer shell electronic configuration and energies are similar and thus give an oxidation state +3. Thus, this results in their similar properties such as similar outer shell electronic configuration, same size, etc.
Do Check Out:





Comments