
Education Journalist | Study Abroad Strategy Lead
Hydrogen is an abundant element found on Earth as well as in outer space. As it resembles halogens and alkali metals, Hydrogen is said to be a unique member of the periodic table.
- It is the lightest element and makes up about 75% of the mass of the universe.
- Hydrogen is a colourless, odourless, and tasteless gas.
- Hydrogen is used as a rocket fuel in industrial processes.
- It is also used to power vehicles or to generate electricity.
- It is extensively used to manufacture ammonia gas to produce fertilizers.
- With oxygen, it produces an oxy-hydrogen flame that is used for welding and cutting processes.
| Table of Content |
Key Terms: Element, Periodic Table, Alkali, Metal, Non-Metal, Gaseous Element, Electron, Proton, Atom, Hydrogen
Uses Of Hydrogen
[Click Here for Sample Questions]
Hydrogen is used in the synthesis of ammonia, methanol, fertilizers, and other chemicals. Some other common applications of hydrogen are –
- It can be used as fuel in cars, trucks, and other vehicles.
- Hydrogen chloride, prepared from hydrogen, is one of the most widely used chemicals.
- Hydrogen fuel cells convert hydrogen and oxygen into electricity and water, with no harmful emissions.
- Hydrogen is used for the process of metallurgical processes to reduce metal oxides.
- The manufacturing of nitrogenous fertilizers happens in the presence of hydrogen
- Hydrogen is mixed with many ingredients to manufacture organic compounds like methanol.
- In many space research activities, hydrogen is utilised as rocket fuel.
- Hydrogen can also be used to generate electricity in power plants.
- It is also used in the refining of petroleum and the production of steel.
- Hydrogen is the primary fuel used in rockets and space shuttles.
- It can also be used to produce hydrogenated oils, which are used in food processing and the production of cosmetics.
Hydrogen
Properties Of Hydrogen
[Click Here for Sample Questions]
Hydrogen has various properties
Physical Properties of Hydrogen
The distinguished physical properties of hydrogen are:
- It is colourless and odourless.
- Hydrogen is lighter than air.
- It is insoluble in water.
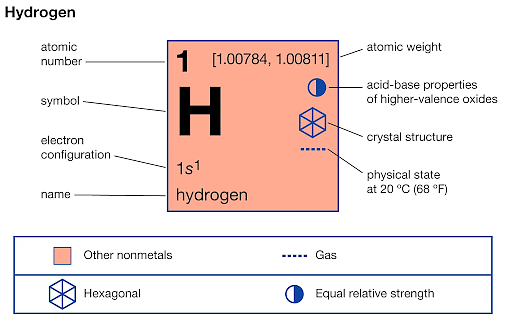
- The atomic number of hydrogen is 1
- The atomic mass of hydrogen is 1.008 amu.
- The density of hydrogen is 0.0899 x 10-3g.cm-3 at 20°C
- The melting point of hydrogen is -259.2 °C
- The boiling point of hydrogen is -252.8 °C
- Hydrogen’s ionization enthalpy is 1312 kJ mol-1.
Chemical Properties of Hydrogen
- Hydrogen has a high bond dissociation enthalpy.
- It requires a lot of energy to break the H-H bond.
- This makes hydrogen relatively inert at room temperature.
- Hydrogen can react by losing an electron to form a cation (H+), or by gaining an electron to form an anion (H-).
- Hydrogen can also share electrons with other elements to form covalent bonds.
Reactions of Hydrogen
[Click Here for Sample Questions]
Hydrogen reacts with other elements to form a variety of compounds.
Halogens
Hydrogen reacts with halogens (fluorine, chlorine, bromine, and iodine) to form hydrogen halides. These reactions release heat (exothermic). The general equation for these reactions is:
H2 + X2 → 2HX, Where X is a halogen.
Oxygen
Hydrogen reacts with oxygen to form water. This reaction is exothermic and also releases light, making it a combustion reaction. The general equation for this reaction is:
2H2 + O2 → 2H2O
Hydrogen and oxygen can react explosively under certain conditions, so it is important to handle these gases with care.
Metals
Hydrogen reacts with many metals to form hydrides. These reactions can be exothermic or endothermic, depending on the metal involved. The general equation for these reactions is:
M + xH2 → MxHx
Where M is a metal and x is the number of hydrogen atoms in the hydride.
Also Read:
| Related Articles | ||
|---|---|---|
| Position of Hydrogen in the Periodic Table | Hydrogen Peroxide | Hydrogen Spectrum |
| Hydrogen | Isotopes of Hydrogen | Hydrogen Bonding |
Things To Remember
- Hydrogen is the lightest and most abundant element in the universe.
- It is odorless, colorless, and tasteless.
- It is a flammable gas and can be used as a fuel or to produce other fuels.
- Hydrogen can be used to generate electricity in power plants and fuel cells.
- It is used in the production of ammonia, fertilizers, methanol, and other chemicals.
- Hydrogen can be used to reduce the sulfur content of fuels, which can help to reduce air pollution.
- It is used in the aerospace industry as fuel for rockets and space shuttles.
Sample Questions
Ques. What is hydrogen? (4 Marks)
Ans. Hydrogen is one of the simplest, tasteless, colourless, odourless, flammable gaseous elements. In hydrogen, each atom has 1 positive electrically charged proton. The element hydrogen is a component composed of many compounds as compared to other elements. It contains three known isotopes.
Hydrogen is an abundantly found element. It is not only found on Earth but also in outer space. Hydrogen is said to be a unique member of the periodic table as it resembles alkali, metals and halogens. Because of its exceptional properties, it has been placed as the first element of the periodic table.
Ques. Name three isotopes of hydrogen. (1 Mark)
Ans. The three isotopes of hydrogen are tritium, deuterium, and protium.
Ques. What is green & brown hydrogen? (2 Marks)
Ans. Green Hydrogen - is when the energy which is used to power the electrolysis comes from renewable sources like wind, water or solar.
Brown Hydrogen - is negligibly environment-friendly hydrogen created through burning coal or lignite. The brown hydrogen creates a lot of carbon dioxide as same as burning the source fuel.
Ques. State the uses of hydrogen. (4 Marks)
Ans. The uses of hydrogen are:
- Hydrogen is used for exploring outer space
- Hydrogen is used in the process of synthesis of ammonia.
- Hydrogen chloride is one of the most important and highly used chemicals in all. This useful chemical is prepared from hydrogen.
- The hydrogen fuel cells and the burning of hydrogen both produce electricity.
- Hydrogen is used for the process of metallurgical processes to reduce metal oxides.
Ques. Does our body require hydrogen? (3 Marks)
Ans. Yes, the human body requires hydrogen, it plays a vital role in keeping our body hydrated. Water is made up of hydrogen and oxygen and it is absorbed by the cells of the body.
Ques. What are the physical properties of hydrogen? (4 Marks)
Ans. The distinguished physical properties of hydrogen are:
- The atomic number of hydrogen is 1
- The atomic mass of hydrogen is 1.007825 g mol-1
- The density of hydrogen is 0.0899*10-3g.cm-3 at 20°C
- The melting point of hydrogen is -259.2 °C
- The boiling point of hydrogen is -252.8 °C
Ques. What are the distinguished methods to prepare hydrogen? (3 Marks)
Ans. There are three different methods for preparing hydrogen.
- Laboratory method
- A commercial preparation method
- Industrial Preparation Of Hydrogen
Ques. What are the atomic number and atomic mass of hydrogen? (1 mark)
Ans. The atomic number of hydrogen is 1 and the atomic mass of hydrogen is 1.007825 g mol-1
Ques. State boiling and melting points of hydrogen. (1 mark)
Ans. The melting point of hydrogen is -259.2 °C and the boiling point of hydrogen is -252.8 °C
Ques. What amounts of hydrogen are required by the human body? (1 mark)
Ans. A human body requires 62.0 % atoms to survive a healthy life.
Ques. What everyday items use hydrogen? (2 marks)
Ans. Some everyday items that use hydrogen are –
- Fertilizer: Hydrogen is used to produce ammonia, which is a key ingredient in many fertilizers.
- Methanol: It produces methanol, which is a fuel and a chemical feedstock.
- Processed foods: Hydrogen is used to hydrogenate fats and oils, which are used in many processed foods.
- Pharmaceuticals: Hydrogen is used in the production of many pharmaceuticals.
- Electronics: Hydrogen is used in the production of semiconductors and other electronic components.
For Latest Updates on Upcoming Board Exams, Click Here:https://t.me/class_10_12_board_updates
Also Check:





Comments