
Education Journalist | Study Abroad Strategy Lead
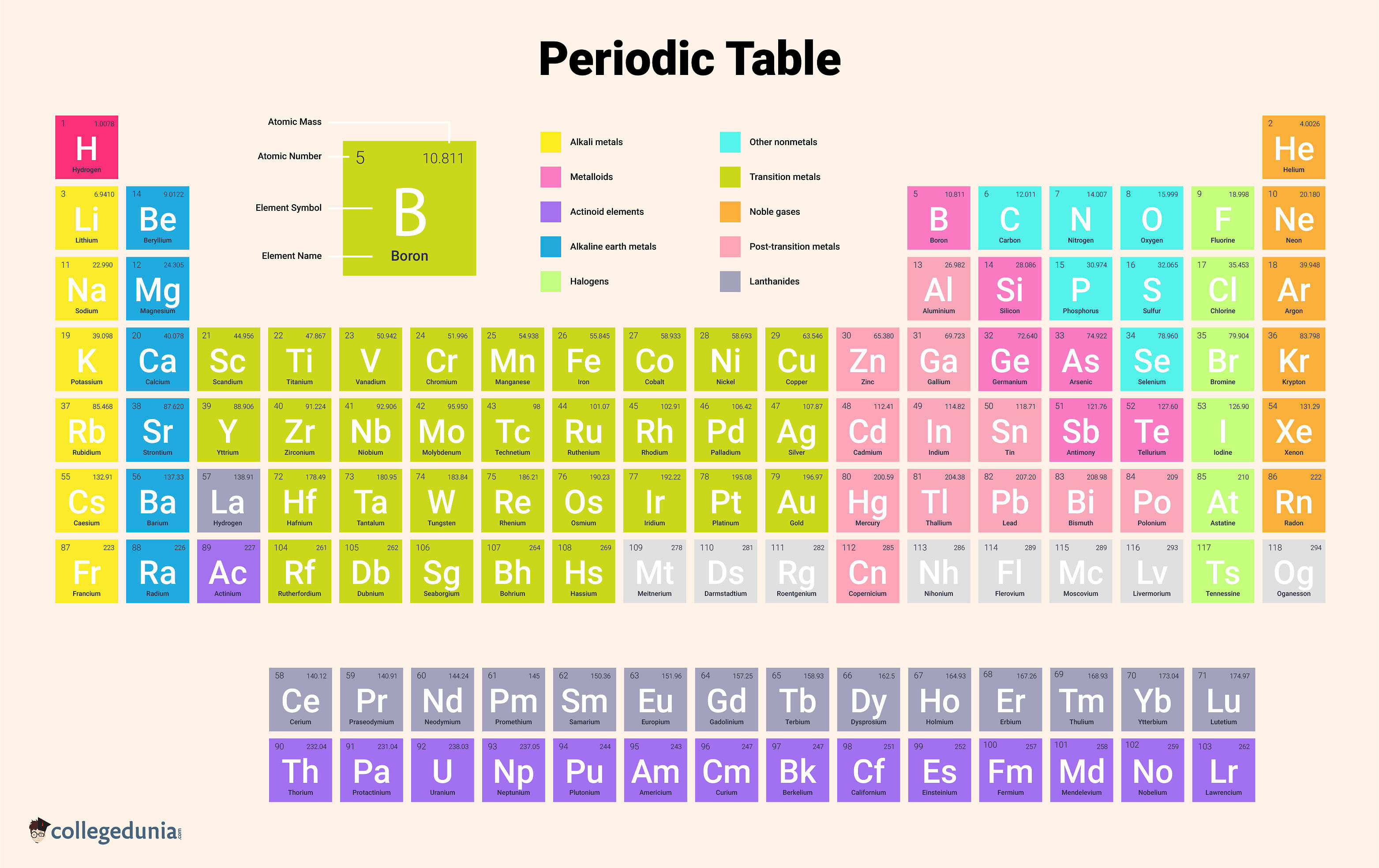
Modern Periodic Table is used in order to organize all the known elements. Elements are arranged in the table based on increasing atomic number, electron setup, and persistent chemical properties. Each element is represented by its chemical symbol in the modern periodic table. The number above each symbol is its atomic number.
- Dmitri Mendeleev is the father of the modern periodic table.
- It is known as the periodic table as the elements are arranged in it in rows and columns.
- The horizontal rows from left to right are known as 'periods' while the vertical columns from up to down are known as 'groups'.
Read More: Periodic Classification of Elements
| Table of Content |
Key Terms: Long Form of Periodic Table, Modern Periodic Table, Mendeleev’s Periodic Table, Electronic Configuration, Atoms, Metals, Non-Metals, Metalloids
Modern Periodic Table
[Click Here for Sample Questions]
Modern Periodic Table is the tabular arrangement of the elements in increasing order of their atomic numbers. It is commonly referred to as the Long Form of the Periodic Table and is based on modern periodic law.
- The elements in the Modern Periodic Table are arranged in a tabular form wherein there are 18 Groups (vertical columns) and 7 Periods (horizontal rows).
- A row is called a period while a column refers to a group.
- In the Modern Periodic Table, the first 94 elements are naturally occurring.
- Whereas the rest of the elements from 95 to 118 have only been synthesized in labs or developed in research centres or atomic reactors.
- The elements in the same group of the Modern Periodic Table have the same valence electron configuration and thus have similar chemical properties.
- The elements in the same period of the Modern Periodic Table will have an increasing order of valence electrons.
- The modern Periodic Table is the modified and improved version of the various arrangement or models developed by various scientists.
- Dimitri Mendeleev developed the periodic table.
- Henry Moseley, an English physicist, perfected the periodic table in 1913, improving on all the flaws of the Mendeleev table.
Modern Periodic Law
[Click Here for Previous Year Questions]
According to the Modern Periodic Law given by Henry Moseley –
The physical and chemical properties of the elements are the periodic functions of their atomic numbers and not their atomic masses.
The elements in the modern periodic table have been arranged in increasing order of their atomic numbers from left to right across each row. And it has been discovered that the elements having similar properties repeat after regular intervals.
Why use atomic number and not atomic mass?Atomic mass is the mass of protons and neutrons present in the nucleus of an atom. While the atomic number is the number of protons in the atom's nucleus. The electrons in the outermost shell of the nucleus are free to move around, so they participate in chemical reactions. Therefore, an element’s properties are dependent on its atomic number and not the atomic mass. |
Read More:
| Other Links Related To This Topic | ||
|---|---|---|
| Group 18 Elements | Group-15 Elements | Group-16 Elements |
| Noble Gases | Dinitrogen | Interhalogen compounds |
Periods in the Modern Periodic Table
[Click Here for Sample Questions]
The rows in the modern or long form of the periodic table are referred to as periods. Each period of the periodic table represents the number of shells or energy levels present in an atom of an element.
- There are a total of 7 periods in the Modern Periodic Table that are numbered as 1, 2, 3, 4, 5, 6 and 7 from top to bottom.
- The number of elements in each period varies. The 1st period has only two elements which are Hydrogen and Helium.
- The 2nd and 3rd period of the table consists of 8 elements each while the 4th and 5th period consists of 18 elements each.
- There are 32 elements in the 6th period of the periodic table and the 7th period also has 32 elements.
- There are four new elements in the 7th period of the periodic table which are 113 (Nihonium), 115 (Moscovium), 117 (Tennessine), and 118 (Oganesson).
- Apart from that, there is a separate panel at the bottom of the long form of the periodic table that consists of 14 elements from the 6th period called the lanthanoids and 14 elements from the 7th period called the actinoids.
Groups in the Modern Periodic Table
[Click Here for Previous Year Questions]
The vertical segments, i.e, the columns in the modern or long form of the periodic table are known as Groups. The elements in a similar group generally show patterns in atomic radii, ionization energy, and electronegativity. The atomic radii of the elements increase from the start to finish in a group.
- There are 18 groups in the long form of the periodic table that are numbered from 1 to 18.
- Each group in the periodic table consists of elements that have the same outer shell electronic configuration.
What is the Cause of Periodicity in the Modern Periodic Table?
The repetition of the similar outer electronic configuration after certain regular intervals is the cause of periodicity in properties.
- Like, for instance, all the elements of Group 1 have a similar outer electronic configuration, ns1, where n refers to the Principal Quantum Number of the outermost shell.
- Similarly, the elements of Group 17 have a similar outer electronic configuration, ns2np5. They are halogens and hence have similar properties to one another.
- Another instance is that the elements of group 18 have an outer electronic configuration of ns2 np6. All these elements have fully filled orbitals and this group comprises non-reactive elements called Inert gases.
Also, we know that the elements in a group in the periodic table have similar properties to the other members of the same group as they all have a similar electron configuration of the outer shell. The periodic table is divided into s, p, d and f blocks on the basis of the entry of the valence electrons in the respective sub-shell.
Classification of Elements in the Long Form of Periodic Table
[Click Here for Sample Questions]
The major classification of elements in the long form of the periodic table or the modern periodic table is described below:
- Alkali and Alkaline Earth Metals: The initial two groups on the left side of the modern periodic table comprise highly reactive elements except for hydrogen. The elements in the first group contain one electron in the valence shell while the elements in the second group contain two electrons in their valence shell.
- Transition Metals: These transition metals are the elements that occupy the centre of the periodic table and display the properties of metals majorly. The transition metals are the elements starting from Group 3 to Group 12. A few transition metals, known as Lanthanides and Actinides, are placed separately in two rows at the bottom of the periodic table.
- Metalloids and Non-Metals: In the periodic table, metalloids appear in a diagonal line on the right side and are basically the elements that separate the metals and nonmetals in the periodic table. Metalloids are those elements that exhibit the properties of both metals and non-metals.
- Noble Gases: Noble Gases occupy the extreme right side of the periodic table and are placed in the 18th Group and have completely filled valence shells. These gases are non-reactive and are also called inert gases.
The study of the elements would have been impossible without the existence of the long form of the periodic table or the modern periodic table as the classification of elements in this way helps in the easier understanding of the properties of elements.
118 Elements and Their Symbols and Atomic Numbers
[Click Here for Previous Year Questions]
The modern periodic table of elements is as follows:
Click here for all the elements of the modern periodic table
| Atomic Number (Z) | Symbol | Element |
|---|---|---|
| 11 | Na | Sodium |
| 12 | Mg | Magnesium |
| 13 | Al | Aluminium |
| 14 | Si | Silicon |
| 15 | P | Phosphorus |
| 16 | S | Sulfur |
| 17 | Cl | Chlorine |
| 18 | Ar | Argon |
| 19 | K | Potassium |
| 20 | Ca | Calcium |
| 21 | Sc | Scandium |
| 22 | Ti | Titanium |
| 23 | V | Vanadium |
| 24 | Cr | Chromium |
| 25 | Mn | Manganese |
| 26 | Fe | Iron |
| 27 | Co | Cobalt |
| 28 | Ni | Nickel |
| 29 | Cu | Copper |
| 30 | Zn | Zinc |
| 31 | Ga | Gallium |
| 32 | Ge | Germanium |
| 33 | As | Arsenic |
| 34 | Se | Selenium |
| 35 | Br | Bromine |
| 36 | Kr | Krypton |
| 37 | Rb | Rubidium |
| 38 | Sr | Strontium |
| 39 | Y | Yttrium |
| 40 | Zr | Zirconium |
| 41 | Nb | Niobium |
| 42 | Mo | Molybdenum |
| 43 | Tc | Technetium |
| 44 | Ru | Ruthenium |
| 45 | Rh | Rhodium |
| 46 | Pd | Palladium |
| 47 | Ag | Silver |
| 48 | Cd | Cadmium |
| 49 | In | Indium |
| 50 | Sn | Tin |
| 51 | Sb | Antimony |
| 52 | Te | Tellurium |
| 53 | I | Iodine |
| 54 | Xe | Xenon |
| 55 | Cs | Caesium |
| 56 | Ba | Barium |
| 57 | La | Lanthanum |
| 58 | Ce | Cerium |
| 59 | Pr | Praseodymium |
| 60 | Nd | Neodymium |
| 61 | Pm | Promethium |
| 62 | Sm | Samarium |
| 63 | Eu | Europium |
| 64 | Gd | Gadolinium |
| 65 | Tb | Terbium |
| 66 | Dy | Dysprosium |
| 67 | Ho | Holmium |
| 68 | Er | Erbium |
| 69 | Tm | Thulium |
| 70 | Yb | Ytterbium |
| 71 | Lu | Lutetium |
| 72 | Hf | Hafnium |
| 73 | Ta | Tantalum |
| 74 | W | Tungsten |
| 75 | Re | Rhenium |
| 76 | Os | Osmium |
| 77 | Ir | Iridium |
| 78 | Pt | Platinum |
| 79 | Au | Gold |
| 80 | Hg | Mercury |
| 81 | Tl | Thallium |
| 82 | Pb | Lead |
| 83 | Bi | Bismuth |
| 84 | Po | Polonium |
| 85 | At | Astatine |
| 86 | Rn | Radon |
| 87 | Fr | Francium |
| 88 | Ra | Radium |
| 89 | Ac | Actinium |
| 90 | Th | Thorium |
| 91 | Pa | Protactinium |
| 92 | U | Uranium |
| 93 | Np | Neptunium |
| 94 | Pu | Plutonium |
| 95 | Am | Americium |
| 96 | Cm | Curium |
| 97 | Bk | Berkelium |
| 98 | Cf | Californium |
| 99 | Es | Einsteinium |
| 100 | Fm | Fermium |
| 101 | Md | Mendelevium |
| 102 | No | Nobelium |
| 103 | Lr | Lawrencium |
| 104 | Rf | Rutherfordium |
| 105 | Db | Dubnium |
| 106 | Sg | Seaborgium |
| 107 | Bh | Bohrium |
| 108 | Hs | Hassium |
| 109 | Mt | Meitnerium |
| 110 | Ds | Darmstadtium |
| 111 | Rg | Roentgenium |
| 112 | Cn | Copernicium |
| 113 | Nh | Nihonium |
| 114 | Fl | Flerovium |
| 115 | Mc | Moscovium |
| 116 | Lv | Livermorium |
| 117 | Ts | Tennessine |
| 118 | Og | Oganesson |
Read More:
Things to Remember
- The Modern Periodic Table is the arrangement of all the known elements as per their increasing atomic number and recurring chemical properties.
- Henry Moseley, in 1913, reformed all the flaws of the Mendeleev table, that was given by Dimitri Mendeleev, the father of the periodic table.
- The Modern Periodic Law governs the Modern Periodic Table which states that the physical and chemical properties of the elements are the periodic functions of their atomic numbers.
- The elements in the periodic table are in a tabular arrangement where a row is called a period and a column refers to a group.
- In the modern periodic table, there are a total of 18 Groups and 7 periods.
- In the Modern Periodic Table, the elements in the same group have the same valence electron configuration and have similar chemical properties.
- The elements in the same period have an increasing order of valence electrons.
Important PYQs Related To This Chapter
- The element with the atomic number 118, will be… (NEET 1996)
- Which of the following element has the highest ionisation enthalpy… (BITSAT 2019)
- A certain compound X when treated with copper… (NEET 1994)
- KF combines with HF to form… (BITSAT 2005)
- Which of the following has zero net dipole moment… (BITSAT 2019)
- In the clathrates of xenon with water…(BITSAT 2019)
- Which one of the following statements is not correct...M(AP EAPCET 2009)
- The chief component of cement is… (JIPMER 2000)
- The molecular electronic configuration of… (BITSAT 2005)
- Although the details of the structure of monoclinic sulphur… (CBSE CLASS XII)
Sample Questions
Ques. What do you understand by the periodic classification of elements? (3 Marks)
Ans. The periodic classification of elements refers to the process of classification by which the elements are grouped on the basis of their properties, that is, the elements that are similar are in one group and the rest of the elements in the other group.
The classification of the elements is now studied with the help of the modern periodic table. The elements have been arranged in the order of their increasing atomic numbers in the long form of the periodic table/modern periodic table. There are seven horizontal rows called periods and 18 vertical columns called groups in the long form of the periodic table.
Ques. Explain how the properties of an element depend on the atomic number rather than the atomic mass. (3 Marks)
Ans. Atomic mass refers to the total mass of the protons and neutrons present in a nucleus of an atom, whereas, the atomic number is defined as the number of protons in a nucleus. One point to be noted here is that the number of protons in the nucleus is equal to the electrons present outside the nucleus.
As we know, since the nucleus is deep-seated inside an atom, but the electrons are outside it, especially the ones in the outermost shell are free to move around. Thus, they take part in chemical reactions, thus, leading to the fact that the properties of an element depend on the atomic number rather than the atomic mass.
Ques. Throw light on the contributions of Mendeleev to the Modern Periodic Table. (3 Marks)
Ans. Dimitri Mendeleev, also known as the father of the periodic table, has given the periodic table that is similar to the modern periodic table which was published in the German Journal of chemistry in 1869. However, there is one aspect in which Mendeleev’s periodic law differs from modern periodic law.
The periodic table formulated by Mendeleev was on the basis of increasing atomic mass, whereas, the modern periodic law is governed by the increasing order of atomic numbers.
At the time of Mendeleev’s Periodic table, only around half of the elements were known and most of the information about the elements was inaccurate. Despite this, Mendeleev was able to predict the discovery and properties of certain elements that were discovered in future later on.
Ques. How many elements are present that are known to man? (3 Marks)
Ans. There are 118 elements that are known to the man currently. In 1800, only 31 elements were discovered, and later on, in 1865, about 63 more elements were discovered.
Of the 118 known elements, there are some elements that are naturally occurring while some are man-made. The first 94 elements of the modern periodic table are naturally occurring, whereas elements from 95 to 118 are synthesized in labs or as nuclear reactors.
Ques. Briefly describe the achievements of Mendeleev's periodic table (3 Marks)
Ans. The major achievements of Mendeleev’s periodic table are as follows:
- He made sure that the elements that had similar properties were placed in the same group, thus, leading to a systematic way to study elements.
- Mendeleev rectified the atomic masses of many elements like gold, platinum and indium, after placing them correctly.
- One of the most significant achievements was the prediction of elements that were yet to be discovered was arranged in the same group. These elements were later on named scandium, germanium and gallium.
- Lastly, Mendeleev placed the noble gases in a new group without disturbing the order that already existed in the table.
Ques. What are Dobereiner’s Triads and what were the limitations of Dobereiner’s classification? (3 Marks)
Ans. In his approach, Dobereiner took three elements with similar properties and grouped them in the order of increasing atomic masses and called this group a triad. Basically with this approach, he showed that the atomic mass of the element present in the middle is the arithmetic mean of the other two elements approximately.
The limitations of Dobreiner’s triad are that Dobereine could find only three triads, i.e, a total of only 9 elements. However, the total number of elements was way more than that of those included in Dobereiner’s Triad. Therefore, he could not classify most of the elements known at that time.
Ques. What is the significance of the Modern Periodic Table? (3 Marks)
Ans. The Modern Periodic Table has made the study of the physical and chemical properties of elements easy by providing us with a fixed pattern in which the elements change their properties periodically.
With the help of the periodic table, one can check or predict the properties of the elements based on their group. Apart from that, scientists can take the help of a periodic table to be aware of the trending characteristics based on the properties of elements and thus be able to identify the new elements with the existing ones.
Ques. What does the Modern Periodic Law imply? (3 Marks)
Ans. The Modern Periodic Law was proposed in 1913 by Henry Moseley, a scientist. As per the Modern Periodic Law, “Properties of elements are the periodic function of their atomic number.” In simpler terms, the law states that the properties of elements depend upon their atomic number and not their atomic masses and the elements are given positions in the periodic table on the basis of their increasing atomic number.
Since the atomic number determines the distribution of electrons in the orbits, the electrons of the outermost orbit determine the properties of an element.
Ques. Why did Mendeleev leave some gaps in the Periodic table? (3 marks)
Ans. Mendeleev left some gaps in the periodic table for yet-to-be-discovered elements. Mendeleev predicted the properties of these elements on the basis of their positions. For example, he predicted the properties of gallium (eka-aluminium) and germanium (eka-silicon) which were unknown then.
Ques. The atomic radii of the first group of elements are given. What is the reason behind the observed trend in the above elements? (2 marks)
| Group-I element | Atomic Radii (pm) |
| Na | 86 |
| K | 231 |
| Rb | 244 |
| Cs | 282 |
Ans. The new shells are being added as we move down the group. This increase the distance between the outermost shell and the nucleus, hence the atomic radii of elements increase gradually down the group in spite of the increase in nuclear change.
Read more:






Comments