
Education Journalist | Study Abroad Strategy Lead
Phosphorus is a mineral that is found in both animal and plant tissue. It can be found in both living and dead beings. Eggs and milk both contain phosphoproteins. Arsenic, antimony, and bismuth are often present in the form of sulphide minerals.
Oxoacids are acids that contain Oxygen components. Phosphorus is known to shape a variety of oxoacids, including H3PO4, H3PO3, and others. It is tetrahedrally surrounded by other atoms in phosphorus oxoacids. Molecular nitrogen makes up 78 percent of the atmosphere's volume.
Read Also: Class 12 Group 15 Elements
| Table of Content |
Key Terms: Interhalogen Compounds, Group 15 Elements, Phosphine, atoms, chemical compound, Boiling Point
Oxoacids of Phosphorus
[Click Here for Sample Questions]
Phosphorus forms a variety of Oxoacids. All these oxoacids form at least one P=O bond and one P–OH bond. The bonds of P–H and P–P are also formed in these acids. Acids with a P–H bond have excellent reducing properties. Hypophosphorous acid is a strong reducing agent since it has two P–H bonds. For example,
4Ag + 4HNO3 + H3PO4 4AgNO3 + 2H2O + H3PO2
These P–H bonds are not ionisable and therefore do not contribute to basicity. Only those H atoms that are bound to oxygen in the atmosphere are considered.
The basicity is caused by the ionisation of the P–OH form. As the structure of H3 PO3 shows, dibasic and tribasic H3PO4 exist. H3PO4 has three P–OH bonds and two P–OH bonds.
Some of the Oxoacids of Phosphorus are given below in the table-

Hypophosphorous Acids (H3PO2)
[Click Here for Sample Questions]
Pierre Louis Dulong, a French chemist, developed hypophosphorous acid for the first time in 1816. The chemical formula for Hypophosphorous is H3PO2 but a more succinct abbreviation is HOP(O)H2, which emphasizes its monoprotic nature. The hypophosphorous Oxidation state of phosphorus is +1. Hypophosphites are the salts made from this acid. Hypophosphorous acid has one P – OH, two P – H and one P=O bond. However, the preparation for the latter is done by white P4+ alkali.

Hypophosphorous Acids (H3PO2)
Preparation of Hypophosphorous Acid
A two-step procedure is used to make the acid in the industrial setting. To begin, hypophosphite salts of alkali and alkaline earth metals are formed when white phosphorus reacts with a hot aqueous solution of the appropriate hydroxide, such as Ba(OH)2.
3Ba( OH)2 + 2P4 + 6H2O → 2PH3 + 3Ba(H2PO2)2
Ba(H2PO2)2 + H2SO4 → BaSO4 + 2H3PO2
The appearance of Hypophosphorous acid is colorless and it has an oily liquid texture.Uses of Hypophosphorous Acid
H3PO2 can be used to reduce arene diazonium salts in organic chemistry, converting ArN+2 to Ar–H. An amine substituent can be extracted from arenes by diazotizing them in a concentrated solution of hypophosphorous acid. While it is mainly used as a salt.
Determination of Basicity of Hypophosphorous Acid
There are two types of bonds in hypophosphorous acid H3PO2: two P-H bonds and one P-OH bond, because only one hydrogen atom is directly bonded to an electronegative oxygen atom. As a result, the basicity of hypophosphorous acid is 1. Basicity sum = 6.
Read More:
| Related Topics | ||
|---|---|---|
| p Block Elements Ncert Solutions | The p-Block Elements Important Questions | P-Block Elements MCQ |
Orthophosphorous Polymerase (H3PO3)
[Click Here for Sample Questions]
It is a potent reducing agent that can quickly reduce several compounds such as copper sulphate to metallic copper, silver nitrate to metallic silver, and iodine to hydriodic acid. It is water-soluble and forms white crystals. It is colorless and odorless in most instances.

Orthophosphorous (H3PO3)
When heated, orthophosphorous decomposes into phosphoric acid and Phosphine. It's made by combining phosphorous trichloride and cold water. It has two P – OH, one P – H, one P = O
Preparation of Orthophosphorous
Orthophosphorous is prepared by the hydrolysis of Phosphorus Trichloride
PCl3 + 3H2O → H3PO3 + 3HCl
One more procedure is used for the preparation of Orthophosphorous. When the Hydrolysis of Phosphorus Trioxide is done then the product that appeared is Orthophosphorous.
P4O6 + 6 H2O → 4H3PO3
Also Read:
Pyrophosphorous Acid (H4P2O5)
[Click Here for Sample Questions]
Pyrophosphorous acid is an obsolete name for the first of two isomeric diphosphonic acids with the formula H4P2O5 and POP and PP bridges. The preparation of Pyrophoric acid requires PCl3 + H3PO. Pyrophosphorous acid has two P – OH, two P–H and two P = O bonds.
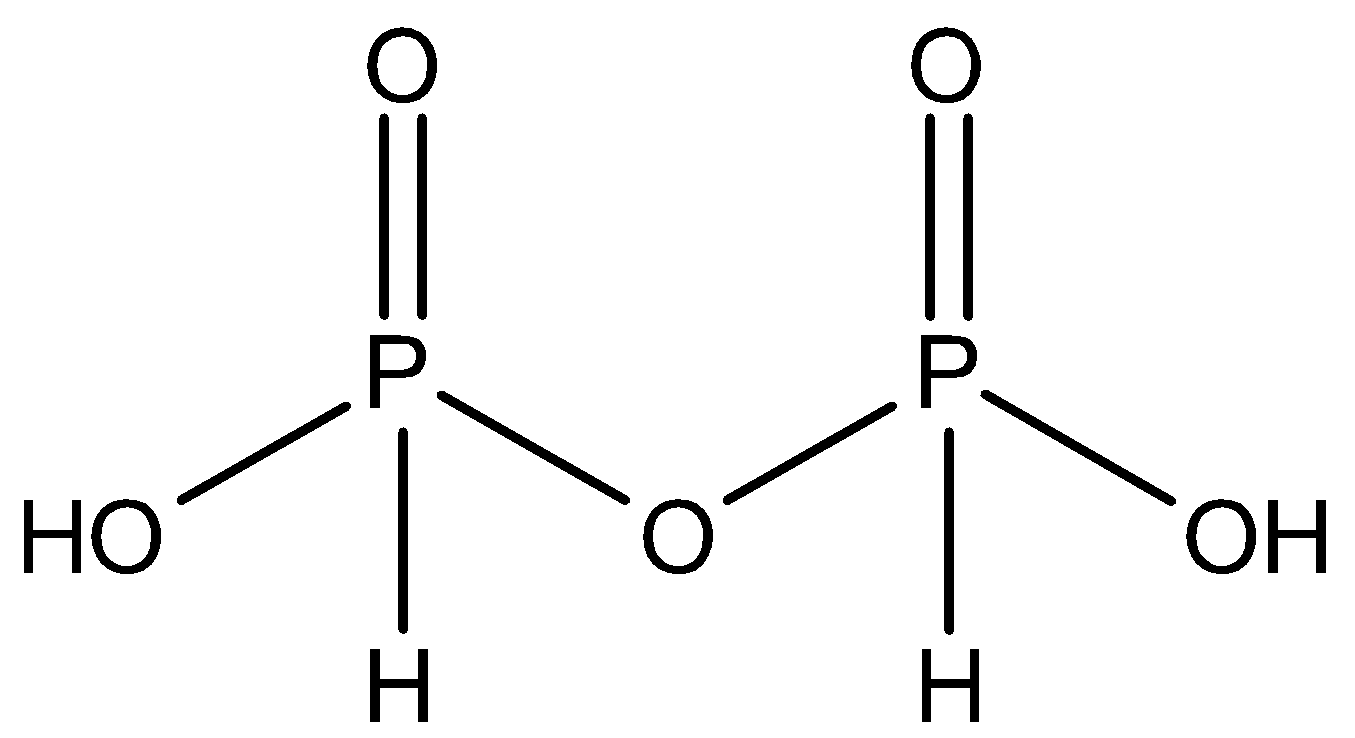
Pyrophosphorous acid (H4P2O5)
The oxidation state of phosphorus for Pyrophosphorous is +3.
Preparation of Pyrophosphorous Acid
Pyrophosphorous Acid is prepared when Orthophosphorous Acid is treated with phosphorus trichloride.
5H3PO3 + PCl3 → 3H4P2O5 + 3HCl
Read More: Aqua Regia
Hypophosphoric Acid (H4P2O6)
[Click Here for Sample Questions]
The mineral acid hypophosphoric acid has the formula H4P2O6, with phosphorus in the formal oxidation state of +4. It exists in solid form as the dihydrate, H4P2O6H2O. The phosphorus atoms in hypophosphoric acid are identical and are joined directly with a PP bond.

Hypophosphoric Acid (H4P2O6)
Preparation of Hypophosphoric Acid
When red phosphorus is reacted with sodium chlorite at room temperature, the hypophosphoric acid is prepared. It is a two step process. Initially Na2H2P2O6 is produced which on further treatment gives H4P2O6 and Sodium. Certainly, the Melting Point for the latter is 54 °C (129 °F; 327 K).
2P + 2NaClO2 + 2H2O → Na2H2P2O6 +2HCl
Na2H2P2O6 + 2H → H4P2O6 + 2Na
Read More: Organometallic Compounds
Orthophosphoric Acid (H3PO4)
[Click Here for Sample Questions]
Orthophosphoric acid, also known as phosphoric(V) acid. The chemical formula of orthophosphoric acid is H3PO4. It is typically found as a colourless syrup with an 85 percent concentration in water. The pure compound is an odourless and colourless solid.

Orthophosphoric acid
When all three H+ ions are removed, the orthophosphate ion PO43-, also classified as "phosphate," is formed. When one or two protons are removed, the dihydrogen phosphate ion H2PO4 and the hydrogen phosphate ion HPO42- are formed respectively. Orthophosphoric acid also produces esters known as organophosphates.
Preparation of Orthophosphoric Acid
Orthophosphoric Acid is prepared when P4O10 is treated with the bubbled water. It is Tribasic in nature.
P4O10 + 6H2O ⇒ 4H3PO4
Uses of Orthophosphoric Acid
Orthophosphoric acid is used in soap and detergents, toothpaste, the food industry, and for water treatment as well. However, all have separate and individual phosphate derivatives specifically.
Read Also: Electron Gain Enthalpy
Metaphosphoric Acid
[Click Here for Sample Questions]
In general, Metaphosphoric acid is a phosphorus oxoacid in which each phosphorus atom is in the oxidation state +5 and is bonded to four oxygen atoms, and one of those is through a double bond and is placed as the corners of a tetrahedron. It has three P – OH, three P = O and three P – O – P bonds.

Metaphosphoric Acid
Preparation of Metaphosphoric Acid
When Pyrophosphoric acid or Orthophosphoric acid are heated till the mixture turns red hot, Metaphosphoric acid is formed.
H4P2O7 → 2HPO3 + H2O
Also when Crystalline phosphorus acid is heated with bromine in a sealed tube, it gives Metaphosphoric acid.
H3PO3 + Br2 → HPO3 + 2HBr
Things to Remember
- Phosphorus is a mineral that is found in both animal and plant tissue.
- Phosphorus forms a variety of Oxoacids which have atleast one P=O bond and one P–OH bond.
- Some of the oxoacids of phosphorus are Hypophosphorous Acids, Orthophosphorus acid, hypophosphoric acid, hypophosphorus acid, metaphophoric acid.
- As only one hydrogen atom is directly bonded to an electronegative oxygen atom, the basicity of hypophosphorous acid is 1.
Read Also: Oxoacids of Sulphur
Sample Questions
Ques. Write the formula of the compound of phosphorus which is obtained when cone. HNO3 oxidises P4. (All India 2017, 1 Mark)
Ans. When Phosphorus is treated with the concentrated HNO? then Phosphoric Acid and Nitrogen dioxide is prepared.
P4 + 20HNO3 → 4H3PO4 + 20NO2 + 4H2O
Ques. What is the basicity of H3PO4? (Delhi 2015, 1 Mark)
Ans. In H3PO4 , there are 3 OH groups present. So the basicity of H3PO4 is 3.

Ques. What is the basicity of H3PO3? (All India 2014, 1 Mark)
Ans. The basicity of H3PO3 is 2. The reason behind the basicity is the number of replaceable H+ ions in an acid. Since H3PO3 is a Dibasic acid so its basicity is 2.
Ques. Draw the structural formulae of the following compounds :
(i) H4P2O5
(ii) XeF4 (All India 2010, 2 Mark)
Ans. (i) H4P2O5 (Pyrophosphorous Acid)

(ii) XeF4

Ques. What happens when
(i) PCl5 is heated?
(ii) H3PO3 is heated? Write the reactions involved. (Delhi 2013, 2 Marks)
Ans. (i) When PCl5 is heated then it decomposes into PCl3 + Cl2
(ii) Orthophosphorous acid on heating gives orthophosphoric acid and phosphine.

Ques. “Orthophosphoric acid (H3PO4) is non-reducing whereas hypophosphorous acid (H3PO2) is a strong reducing agent.” Explain and justify the above statement with suitable examples. (Comptt. Delhi 2017, 2 Marks)
Ans. Orthophosphoric acid (H3PO4) does not contain any P-H bond whereas hypophosphorous acid (H3PO2) contains two P-H bonds. That is why Orthophosphoric acid (H3PO4) is non-reducing whereas hypophosphorous acid (H3PO2) is a strong reducing agent.
For Example- H3PO2 can reduce silver nitrate (AgNO3) into metallic silver but H3PO4 can not.
4AgNO3 + H3PO2 + 2H2O → 4Ag ↓ + H3PO4 + 4HNO3
Ques. Give reasons for the following :
(i) (CH3)3 P = O exists but (CH3)3 N = O does not.
(ii) Oxygen has less electron gain enthalpy with negative sign than sulphur.
(iii) H3PO2 is a stronger reducing agent than H3PO3. (All India 2014, 3 Marks)
Ans. (i) (CH3)3P = 0 exists due to the presence of an empty d-orbitals and thus can expand its covalency upto 6 on the other hand (CH3)3 N = O cannot expand its covalency due to absence of d-orbitals that is why it does not exists.
(ii) The least negative electron gain enthalpy of oxygen is due to its smaller size and more interelectronic repulsion with the coming electron.
(iii) H3PO2 contains two P-H bonds while H3PO3 contains only one P-H bond. That is why H3PO2 is a stronger reducing agent than H3PO3.
Ques. (i) Ammonia is more basic than phosphine.
(ii) Elements of Group 16 generally show lower value of first ionisation enthalpy compared to the elements in the corresponding periods of Group 15. (All India 2011, 3 Marks)
Ans. (i) Ammonia is more basic than phosphine because Nitrogen is smaller in size than Phosphorus and lone pairs of electrons on Nitrogen are readily available for protonation.
(ii) Elements of Group 16 generally show lower value of first ionisation enthalpy compared to the elements in the corresponding periods of Group 15 because group 15 elements have half filled p-orbitals which are more stable.
Ques. What is the melting and boiling point of Hypophosphorous acid?(1 mark)
Ans. The melting point is 26.5 °C (79.7 °F; 299.6 K) and the boiling point is 130 °C (266 °F; 403 K).
Ques. Based on its structure, how do you account for H3PO2's reducing behavior?(1 mark)
Ans. Two H atoms are directly bonded to a P atom in H3PO2, giving the acid a reducing character.
Ques. What happens when you heat H3PO3?(1 mark)
Ans. H3PO3 decomposes into phosphoric acid and phosphine when exposed to heat.
Ques. What is the preparation for Hypophosphoric acid?(1 mark)
Ans. The preparation for Hypophosphoric acid is red P4 + alkali.
2P + 2NaClO2 + 2H2O → Na2H2P2O6 + 2HCl
Na2H2P2O6 + 2H → H4P2O6 + 2Na
Ques. What are the Characteristic bonds and their number for Orthophosphoric acid?(2 marks)
Ans. Orthophosphoric acid has three P – OH and One P = O bond.

For Latest Updates on Upcoming Board Exams, Click Here: https://t.me/class_10_12_board_updates
Check-Out:





Comments